THE AIMS
Evidence Based Medicine for students
Dr. Khalid AlQumaizi
By the end of this session the student will be able to:
- Know what is evidence-based medicine.
- Identify the main steps of EBM.
- Formulate an answerable question.
- Know common EBM jargons definitions.
- Know hierarchy of evidence.
- Understand the appraisal checklist of article about RCT.
- Understand the appraisal checklist of article about diagnostic test.
- Understand the appraisal checklist of article about Systemic Review.
WHAT IS EVIDENCE-BASED MEDICINE?
Evidence-based medicine (EBM) is the application of current, best-available clinical evidence to health care decisions for individual patients.
WHAT ARE THE KEY STEPS OF EBM? Z
EBM consists of five major steps:
- Asking a clinical question.
- Systematic retrieval of the best-available evidence.
- Critical appraisal of the evidence.
- Application of the evidence.
- Evaluation of performance.
HOW TO FORMULATE AN ANSWERABLE QUESTION
- P.I.C.O
- P - population
- I - intervention
- C - control/comparison
- O - outcome/s
COMMON EBM JARGONS DEFINITIONS
Randomised Controlled Trial (RCT)
- Clinical trial where at least two treatment groups are compared. One must be a control group, e.g., receiving standard care or a placebo treatment. Allocation to a group must be random and unbiased.
Randomisation
- Process of allocating individuals to the alternative treatments in a clinical trial, avoiding bias. Should produce groups which are similar, except for the treatment of interest.
Blinding
- The process of ensuring that participants or researchers (single-blind) or participants and researchers (double-blind) are unaware of which treatment group participants have been randomised to, reducing the possibility of bias in the results.
Intention-to-treat analysis (ITT)
- All patients allocated to one arm of a RCT are analysed in that arm, whether or not they completed the prescribed treatment/regimen.
Additional Jargon Definitions
-
Experimental event rate (EER)
Risk (or chance) of outcome event in experimental group. -
Control event rate (CER)
Risk (or chance) of outcome event in control group. -
Relative risk (RR)
A measure of the chance of the event occurring in the experimental group relative to it occurring in the control group. -
Relative risk reduction (RRR) Z The difference in the proportion of events between the control and experimental groups, relative to the proportion of events in the control group. Can also be calculated as 1-RR.
-
Absolute risk reduction (ARR) Z The absolute difference between the risk of the event in the control and experimental groups.
-
Number needed to treat (NNT) Z The number of patients who needed to be treated to prevent the occurrence of one adverse event (e.g. complication, death) or promote the occurrence of one beneficial event (e.g. cessation of smoking).
-
Confidence interval
For whatever effect being measured (e.g. RR, RRR, ARR, NNT) the confidence interval is the range of values within which the “true” value in the population is found. Generally expressed as a 95% confidence interval, i.e. you can be 95% confident that the population value lies within those limits. -
Cross-sectional study
A study that examines the relationship between disease (or other health-related characteristics) and other variables of interest as they exist in a defined population at a single point in time. -
Sensitivity
The proportion (fraction) of those people who really have the disease (a+c) who are correctly identified as such (a), i.e. the true positives. -
Specificity
The proportion (fraction) of those people who really do not have the disease (b+d) who are correctly identified as such (d), i.e. the true negatives. -
Positive predictive value (PPV)
The proportion (fraction) of the people who test positive (a+b) who truly have the disease (a). -
Negative predictive value (NPV)
The proportion of people who test negative (p+d) who truly do not have the disease (d). -
Pre-test probability
The probability (chance) of a patient having the disease before the diagnostic test is carried out. This is the same as the prevalence of that disease in a population similar to the patient. It may be estimated from routine data, practice data or clinical judgement. -
Post-test probability
After running the diagnostic test, the post-test probability of the patient having the disease is the number of people who truly have the disease (a) as a proportion of those who tested positive (a+b). This is the same as the positive predictive value. -
Systematic review
A review in which evidence on a topic or research question has been systematically identified, appraised and summarised according to predetermined criteria. Systematic reviews may incorporate meta-analysis, but don’t have to. -
Meta-analysis
A statistical technique. Summarises the results of several studies into a single estimate, giving more weight to larger studies. -
Publication bias
When only studies with positive results are published, not the neutral or negative studies. If only published studies are included in a systematic review, it may overestimate the effect of the treatment or intervention.
HIERARCHY OF EVIDENCE
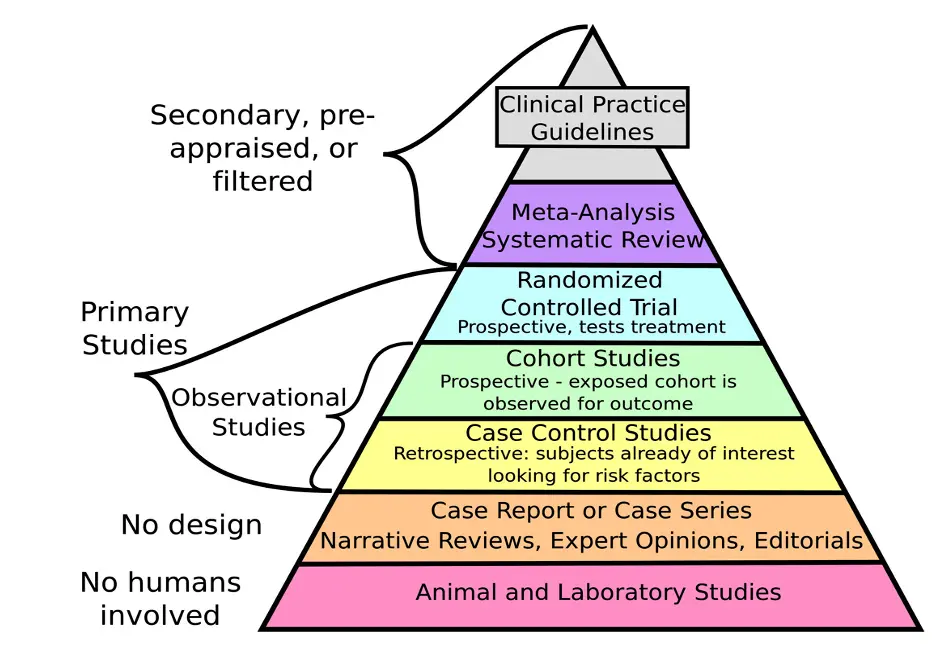
RANDOMISED CONTROLLED TRIALS (RCT)
The Questions
-
Were the following clearly stated:
- Patients
- Intervention
- Comparison Intervention
- Outcome(s)
-
Was the assignment of patients to treatments randomized?
-
Was the randomisation list concealed?
-
Were all subjects who entered the trial accounted for at its conclusion?
-
Were they analysed in the groups to which they were randomised, i.e. intention-to-treat analysis?
-
Were subjects and clinicians ‘blind’ to which treatment was being received?
-
Aside from the experimental treatment, were the groups treated equally?
-
Were the groups similar at the start of the trial?
-
How large was the treatment effect?
-
. Consider how were the results expressed (RRR, NNT, etc).
-
How precise were the results?
-
. Were the results presented with confidence intervals?
-
Do these results apply to my patient?
- Is my patient so different from those in the trial that results don’t apply?
- How great would the benefit of therapy be for my particular patient?
- Are my patient’s values and preferences satisfied by the intervention offered?
- Do I have a clear assessment of my patient’s values and preferences?
- Are they met by this regimen and its potential consequences?
DIAGNOSTIC TEST
The Questions
-
Is this test relevant to my practice?
-
Was there an independent, blind comparison with a reference (‘gold’) standard of diagnosis?
-
Was the diagnostic test evaluated in an appropriate spectrum of subjects (like those to whom it would be offered in practice)?
-
Was the reference standard applied regardless of the diagnostic test result?
-
What were the results?
Consider how were the results expressed (sensitivity, specificity, likelihood ratios)? -
How precise were the results?
-
- Were the results presented with a range or confidence intervals?
-
Were likelihood ratios for the test results presented or data necessary for their calculation provided?
-
Will the test be available, affordable, accurate and reliable in my setting?
-
- Consider: Are the patients similar to my patient? Is the setting similar to my own?
-
If the test isn’t available, were the methods for performing the test described in sufficient detail to permit replication?
-
Can you generate a clinically sensible estimate of your patient’s pre-test probability (e.g. from practice data, clinical judgement or the report itself)?
-
Will the resulting post-test probabilities affect your management and help your patient? (Could it move you across a test-treatment threshold?)
-
Would the consequences of the test help this patient?
SYSTEMATIC REVIEW
Did the review address a clearly focussed issue? Was there enough information on: • The population studied • The intervention given • The outcomes considered 1
Did the authors look for the appropriate sort of papers? The ‘best sort of studies’ would • Address the review’s question • Have an appropriate study design 2
Do you think the important, relevant studies were included? Look for • Which bibliographic databases were used • Follow up from reference lists • Personal contact with experts • Search for unpublished as well as published studies • Search for non-English language studies 3
Did the review’s authors do enough to assess the quality of the included studies? The authors need to consider the rigour of the studies they have identified. Lack of rigour may affect the studies results. 4
If the results of the review have been combined, was it reasonable to do so? Consider whether • The results were similar from study to study • The results of all the included studies are clearly displayed • The results of the different studies are similar • The reasons for any variations are discussed 5
What is the overall result of the review? Consider • If you are clear about the reviews ‘bottom line’results • What these are (numerically if appropriate) • How were the results expressed (NNT, odds ratio, etc) 6
How precise are the results? Are the results presented with confidence intervals? 7
Can the results be applied to the local population? Consider whether • The patients covered by the review could be sufficiently different from your population to cause concern • Your local setting is likely to differ much from that of the review 8
Were all important outcomes considered? 9
Are the benefits worth the harms and costs? Even if this is not addressed by the review, what do you think? 10