Iron Toxicity
Fahad Abuguyan
Overview
Iron is essential to the function of hemoglobin, myoglobin, many cytochromes, and many catalytic enzymes, but becomes extremely toxic when levels are elevated after an overdose or from accumulation in disease states. Iron poisoning used to be the leading cause of poisoning death in children.
Key Points
- The acute ingestion of iron is especially hazardous to children
- Serious iron ingestions in adults are usually associated with suicide attempts
- Acute iron poisoning can result in gastrointestinal symptoms, metabolic acidosis, and hepatotoxicity
Pharmacokinetics and Absorption
- Iron is absorbed in the small intestine
- As little as 10% or as much as 95% of the ingested iron is taken into the cell
- In the cell, iron has three fates: storage bound to ferritin, transfer to the serum where it is bound to transferrin, or loss when the intestinal cell is sloughed off
- Under normal conditions only 15% to 35% of the iron-binding capacity of transferrin is used
Normal Laboratory Values
- Normal serum iron concentrations: 50 to 150 μg/dL
- Total iron-binding capacity (TIBC): 300 to 400 μg/dL (crude measure of serum proteins’ ability to bind iron)
- When iron concentrations rise after a significant overdose, transferrin becomes saturated so that excess iron circulates free and unbound in the serum
- Unbound iron is directly toxic to target organs
Mechanism of Toxicity
Iron causes toxicity through several mechanisms, including:
- Cellular reactions with formation of free radicals and oxidative damage
- Metabolic alterations - changes cellular energy and metabolism
- Mitochondrial dysfunction - uncouples oxidative phosphorylation
- Cellular death - ultimate result of the above mechanisms
Risk Assessment
Risk assessment is based on the amount of elemental iron ingested. The total amount of elemental iron ingested can be approximated by multiplying the estimated number of tablets by the fraction of elemental iron contained in the tablet.
Common Iron Preparations
| COMPOUND | PERCENTAGE OF ELEMENTAL IRON |
|---|---|
| IONIC COMPOUNDS | |
| Ferrous sulfate | 20% |
| Ferrous fumarate | 33% |
| Ferrous gluconate | 12% |
| NON-IONIC COMPOUNDS | |
| Carbonyl iron | 100% |
| Iron polysaccharide | 46% |
Clinical Example
Case: 30 year-old female (wt = 60kg) presents with a history of ingestion of 90 tablets of ferrous sulfate 325 mg tablet
Calculation: Iron equivalents can be determined as below:
(Strength × % elemental Fe/tablet) × # of tablets/weight (kg)
(325 mg × 20% elemental Fe/tablet) × 90 tablets/60 kg = **97.5 mg/kg**
Pathophysiology
Iron has local gastrointestinal effects followed by systemic effects (that do not occur without preceding GI toxicity following iron ingestion).
Local Effects
Corrosive injury to the gastrointestinal mucosa resulting in:
- Vomiting and diarrhea
- Hematemesis and melena
- Fluid losses that may result in hypovolemia
Systemic Effects
Iron acts as a cellular toxin targeting:
- Cardiovascular system and liver (primary targets)
- Central nervous system (secondary effects)
- Metabolic acidosis and coagulopathy
Dose-Response Relationship
| Elemental Iron Dose | Clinical Manifestations | Prognosis |
|---|---|---|
| <20 mg/kg | Asymptomatic | Benign |
| 20-60 mg/kg | Mild to moderate symptoms | Generally recoverable |
| 60-120 mg/kg | Severe morbidity and mortality | Poor prognosis |
| >120 mg/kg | Potentially lethal | Life-threatening |
Additional Critical Doses:
- 50% mortality dose: 200 to 250 mg/kg in adults
- Lethal dose in children: as small as 130 mg of elemental iron
Clinical Features
The clinical effects of acute iron poisoning occur in five distinct stages, though not every patient goes through every phase.
Clinical Manifestations by Phase
| Phase | Timeframe | Clinical Features | Mechanism of Toxicity |
|---|---|---|---|
| 1 | Gastrointestinal (≤6 hours) | • Vomiting • Diarrhea • Hematemesis • Hematochezia | Corrosive effect of iron on gastrointestinal mucosa |
| 2 | Latent (6-24 hours) | • Resolution of GI symptoms • Tachycardia • Acidosis • Depressed mental status | Ongoing cellular toxicity and organ damage |
| 3 | Systemic (12-24 hours) | • Return of GI symptoms • Metabolic acidosis • Leukocytosis • Coagulopathy • Renal failure • Lethargy or coma • Cardiovascular collapse | Iron distributes to tissues with worsening cellular toxicity |
| 4 | Hepatic (2-5 days) | • Fulminant liver failure • Coagulopathy | Rapid absorption from portal system with oxidative damage |
| 5 | Obstructive (3-6 weeks) | • Pyloric or bowel scarring • Obstruction | Healing of injured gastrointestinal mucosa |
Key Clinical Indicators
- No vomiting within 6 hours after ingestion → likely non-toxic dose
- Serum iron levels may fall to normal during phase 3 due to tissue distribution
Diagnosis
History
Critical historical elements include:
- Amount and type of iron preparation ingested
- Time of ingestion
- Presence of vomiting (key prognostic indicator)
Laboratory Evaluation
Essential Laboratory Tests
- Complete Blood Count (CBC) - assess for anemia, leukocytosis
- Renal profile and electrolytes - evaluate renal function
- Liver Function Tests (LFTs) - monitor for hepatic toxicity
- Arterial/Venous Blood Gas - assess for metabolic acidosis
- Serum glucose - hypoglycemia can occur
- Coagulation studies - PT/INR, PTT for coagulopathy
- Serum iron level - definitive diagnostic test
Serum Iron Concentration
- Peak levels occur 4-6 hours following acute iron ingestion
- Levels usually correlate with severity of toxicity initially
- After 8-12 hours iron levels fall due to intracellular shift
- Levels may not clearly correlate with clinical toxicity in later phases
Predicted Toxicity Based on Laboratory Values
| Predicted Clinical Effects | Elemental Iron Dose | Serum Iron Concentration |
|---|---|---|
| Nontoxic or mild GI symptoms | <20 mg/kg | <300 μg/dL (<54 μmol/L) |
| Significant GI symptoms ± systemic toxicity | 20-60 mg/kg | 300-500 μg/dL (54-90 μmol/L) |
| Moderate to severe systemic toxicity | >60 mg/kg | >500 μg/dL (>90 μmol/L) |
| Severe systemic toxicity | >60 mg/kg- | >1000 μg/dL (>180 μmol/L) |
Important: Because iron is rapidly cleared from serum and deposited in liver, concentrations may be deceptively low if measured many hours after peak absorption.
Blood Gas Analysis
- High anion-gap metabolic acidosis is a useful marker of systemic toxicity
- In the absence of iron levels, serum bicarbonate can serve as a surrogate marker
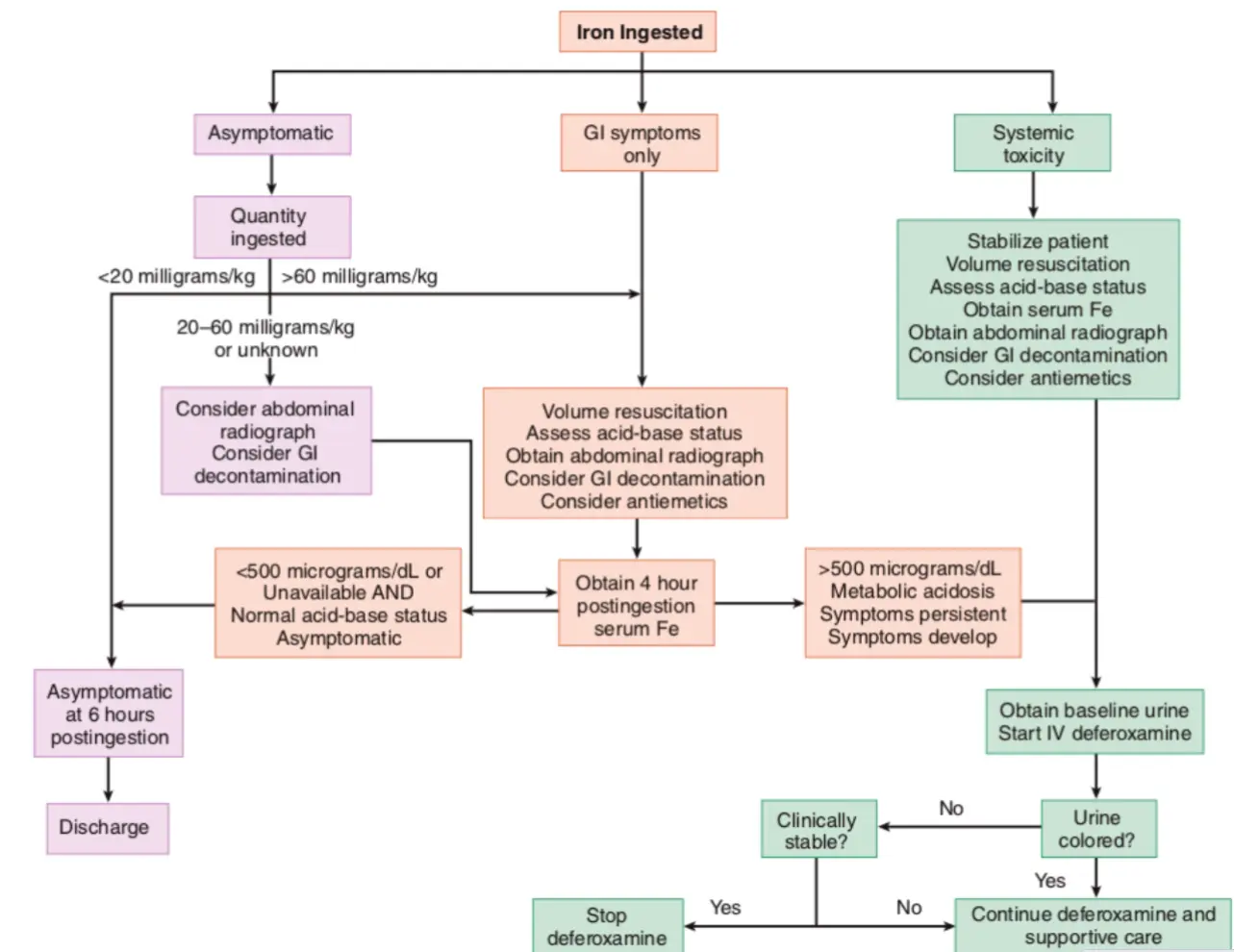
Management
Supportive Measures
- ABCs - Airway, Breathing, Circulation management
- Early volume restoration is priority due to potential GI fluid losses
- Monitor vital signs closely, especially blood pressure
Decontamination
- Activated charcoal: Not effective for iron (iron is not adsorbed)
- Whole-bowel irrigation: Generally the preferred method for significant iron ingestions
- Hemodialysis and hemoperfusion: Not effective due to iron’s large volume of distribution
Antidote Therapy: Deferoxamine
Deferoxamine is the antidote of choice for serious iron overdose.
Indications for Deferoxamine
- Severe clinical symptoms
- High anion-gap metabolic acidosis
- Serum iron level > 500 μg/dL
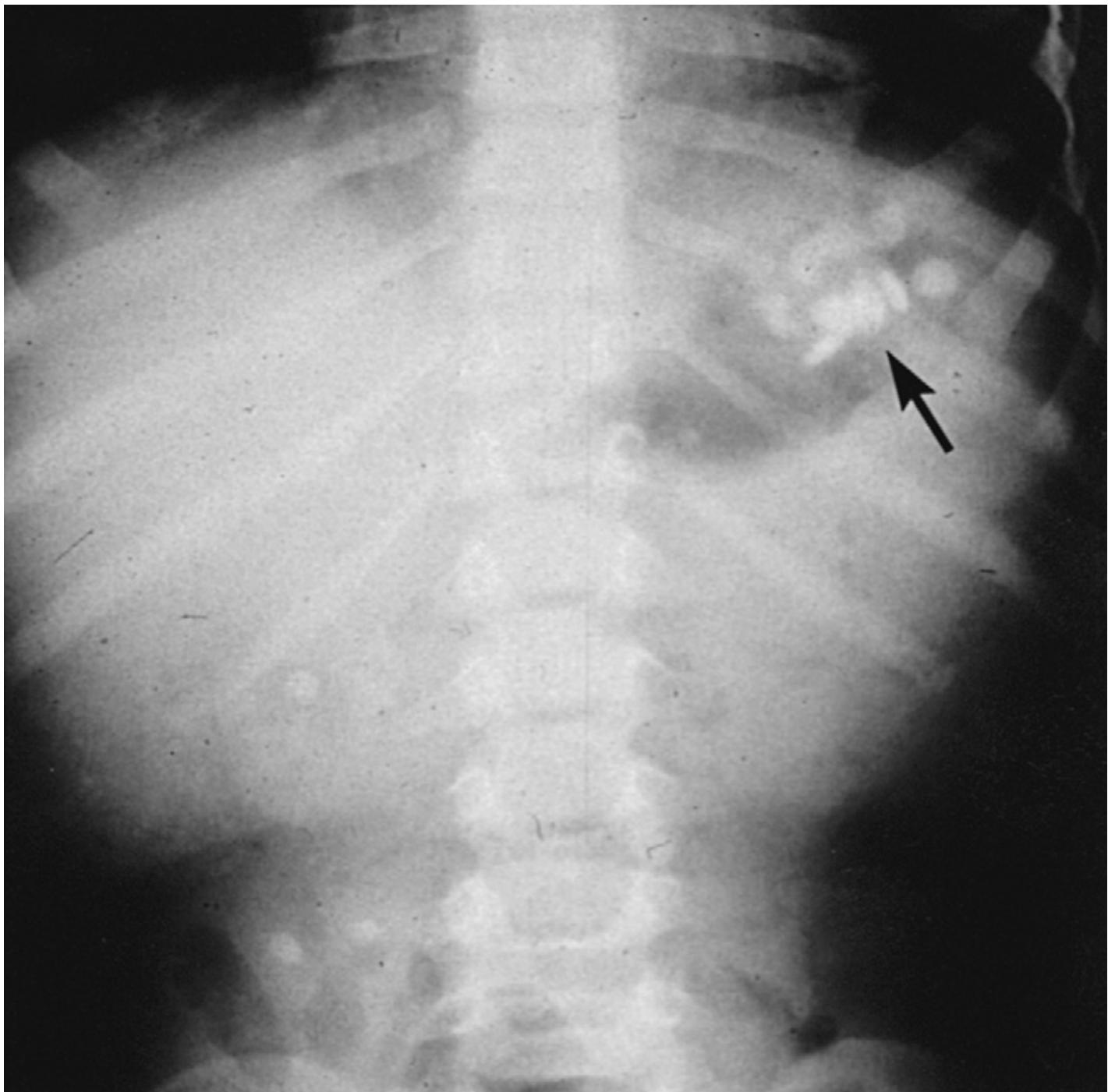
- Significant number of pills visible on abdominal radiography
Dosing and Administration
- Continuous IV infusion at 15 mg/kg/hr for up to 24 hours
- Maximum administration rate: 35 mg/kg/hr
Adverse Effects
- Hypotension (most common significant adverse effect)
- Acute respiratory distress syndrome (ARDS)
- Pulmonary toxicity with prolonged therapy
Clinical tip: Monitor urine color - “vin rose” or orange-red discoloration indicates iron-deferoxamine complex formation and therapeutic effect
Disposition Criteria
Low-Risk Patients
The asymptomatic patient who has reliably ingested < 40 mg/kg of elemental iron does not need additional therapy after observation.
Moderate-Risk Patients (≥ 40 mg/kg ingestion)
Serial iron concentrations should be obtained:
- 3-5 hours post-ingestion
- 6-8 hours post-ingestion
Discharge criteria: If peak iron remains < 300 μg/dL, is not rising, and the patient remains asymptomatic during 6 hours of observation, the patient can be discharged home.
Clinical Management Algorithm