Electrolytes Imbalance
Dr. Eatimad Osheik
s p p c m
Definition – Electrolyte Imbalance
Electrolyte imbalance refers to an abnormality in the concentration of serum electrolytes that disrupts normal physiological function.
potassium calcium ±
Major Electrolytes
- Sodium (Na⁺)
- Potassium (K⁺)
- Calcium (Ca²⁺)
- Magnesium (Mg²⁺)
- Chloride (Cl⁻)
- Bicarbonate (HCO₃⁻)
Normal Role
- Maintain fluid balance
- Regulate acid-base status
- Enable nerve conduction
- Support muscle contraction (including cardiac muscle)
- Maintain cellular homeostasis
Causes of Imbalance
Imbalance may occur due to:
- Renal disorders
- Gastrointestinal losses
- Endocrine diseases
- Medications
- Critical illness
Electrolyte disturbances are common, life-threatening, and reversible causes of morbidity and mortality.
Why It Is Important
- Can cause cardiac arrhythmias (especially potassium imbalance)
- Can lead to seizures and coma (severe sodium disorders)
- Common in hospitalised and ICU patients
- Frequently seen in:
- Chronic kidney disease
- Heart failure
- Diabetes mellitus
- Influences drug safety (e.g., digoxin, diuretics)
Potassium Imbalance
Physiology of Potassium
- Normal serum K⁺: 3.5–5.0 mmol/L
- 98% intracellular (mainly muscle)
- Major determinant of resting membrane potential
- Small changes → major cardiac effects
Regulation of Potassium
-
Renal excretion (most important)
- Distal nephron
- Influenced by:
- Aldosterone
- Sodium delivery
- Acid-base status
- Flow rate
-
Transcellular shifts
- Insulin → shifts K⁺ into cells
- β2 stimulation → shifts K⁺ into cells
- Acidosis → shifts K⁺ out of cells
- Alkalosis → shifts K⁺ into cells
Hypokalaemia (K⁺ <3.5 mmol/L)
Severity
| Severity | Range |
|---|---|
| Mild | 3.0–3.5 mmol/L |
| Moderate | 2.5–3.0 mmol/L |
| Severe | <2.5 mmol/L |
Causes of Hypokalaemia
1. Reduced Intake
- Rare alone
2. Transcellular Shift
- Insulin therapy
- β2 agonists
- Alkalosis
- Refeeding syndrome
3. Increased Loss (Most Common)
A. Gastrointestinal Loss:
- Vomiting
- Diarrhoea
- NG suction
Vomiting causes:
- Metabolic alkalosis
- Secondary hyperaldosteronism
B. Renal Loss:
Diuretics (commonest cause):
- Loop
- Thiazides
Hyperaldosteronism:
- Primary: Conn’s syndrome
- Secondary: Renal artery stenosis
Clinical Features of Hypokalaemia
Neuromuscular:
- Weakness
- Cramps
- Paralysis (severe)
Cardiac:
- Arrhythmias
- Digoxin toxicity risk
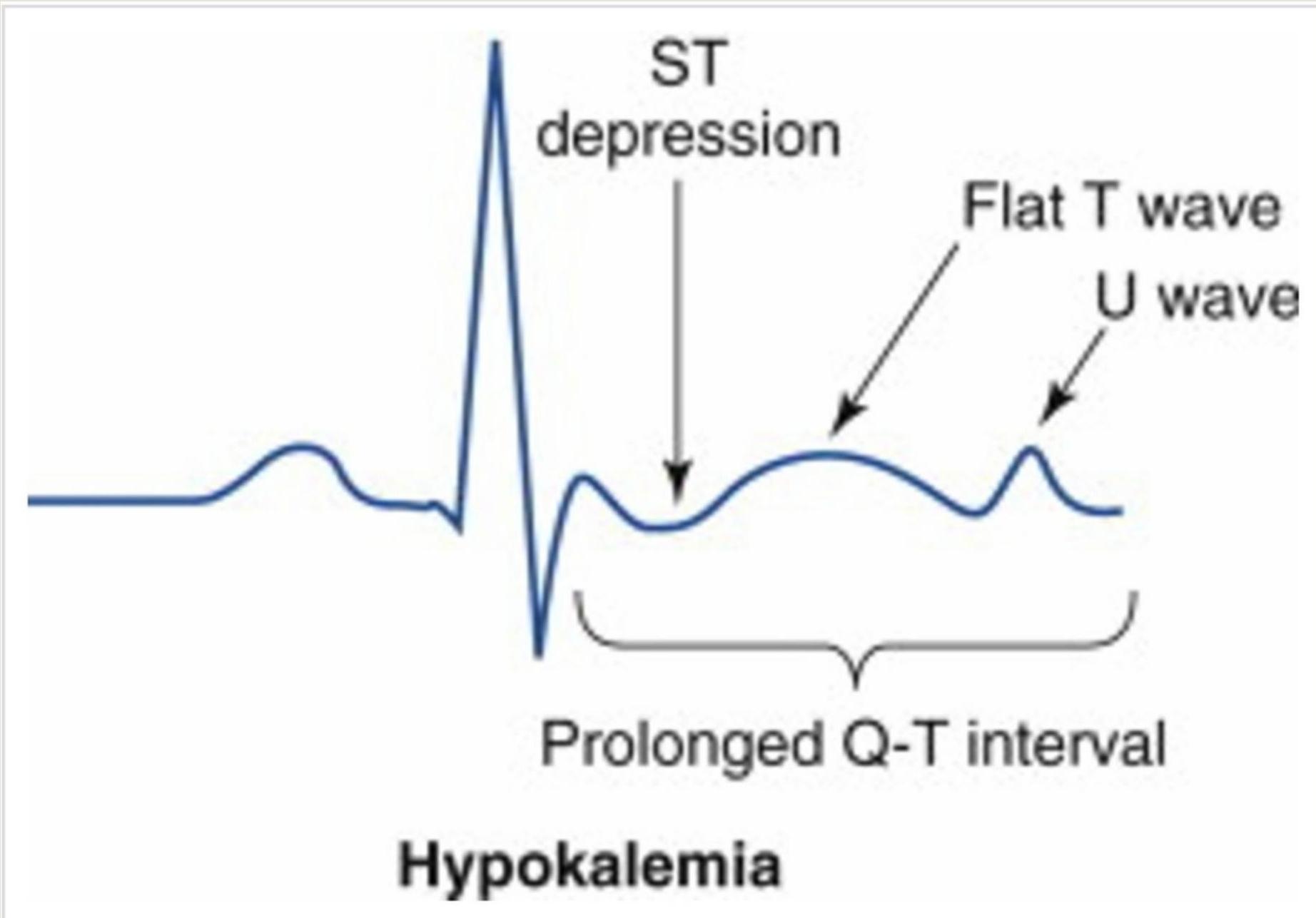
ECG Changes in Hypokalaemia
- Flattened T waves
- Prominent U waves
- ST depression
- Prolonged QT
Evaluation Approach
- Confirm true value
- Check acid-base status
- Measure urine potassium
Urine K⁺:
- <20 mmol/day → extrarenal loss
- >20 mmol/day → renal loss
Management of Hypokalaemia
Mild (asymptomatic):
- Oral potassium chloride
Severe or symptomatic:
- IV potassium (careful!)
IV Rules:
- Max 10 mmol/hr (peripheral)
- Continuous ECG monitoring
- Never give as bolus
Correct magnesium if low
Hyperkalaemia (K⁺ >5.0 mmol/L)
Severity
| Severity | Range |
|---|---|
| Mild | 5.0–5.5 mmol/L |
| Moderate | 5.5–6.5 mmol/L |
| Severe | >6.5 mmol/L |
Causes of Hyperkalaemia
1. Pseudohyperkalaemia
Always exclude first:
- Haemolysis
- Thrombocytosis
- Difficult venepuncture
2. Reduced Renal Excretion (Most Common)
- Acute or Chronic Kidney Disease
- Hypoaldosteronism
- Addison’s disease
- ACE inhibitors
- ARBs
- Potassium-sparing diuretics
3. Transcellular Shift
- Acidosis
- Insulin deficiency (DKA)
- Tissue breakdown (rhabdomyolysis)
- Tumour lysis syndrome
4. Excess Intake
- Rare unless renal impairment
Clinical Features of Hyperkalaemia
- Muscle weakness
- Paraesthesia
- Life-threatening arrhythmias
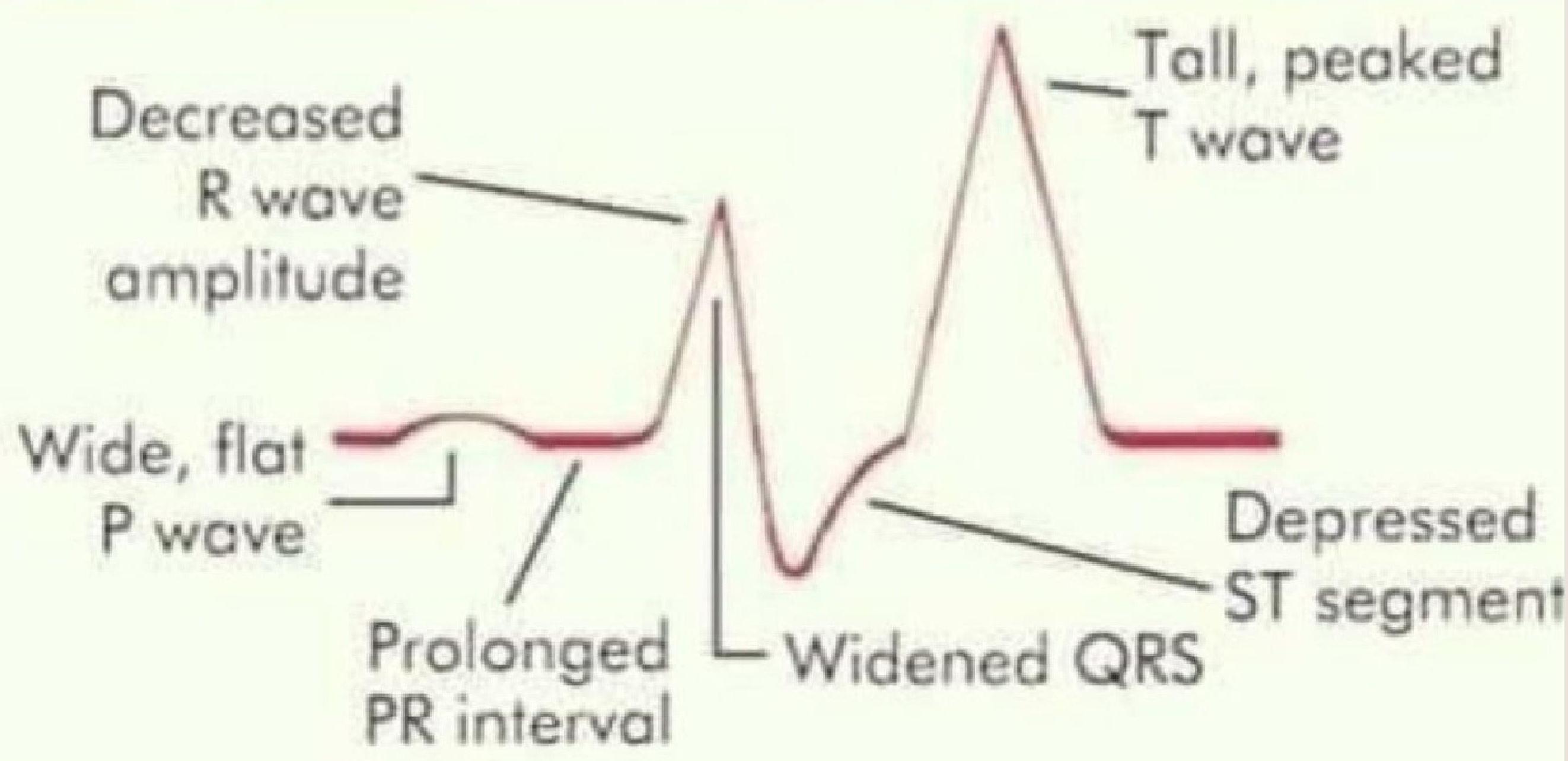
ECG Changes in Hyperkalaemia
Progressive pattern:
- Tall peaked T waves
- Shortened QT
- Prolonged PR
- Wide QRS
- Sine wave pattern
- Ventricular fibrillation/asystole
ECG changes do not always correlate with potassium level.
Emergency Management of Hyperkalaemia
Indications for urgent treatment:
- K⁺ ≥6.5 mmol/L
- ECG changes
- Rapid rise
Stepwise Emergency Management:
1. Stabilize Cardiac Membrane
- IV Calcium gluconate
- Acts within minutes
- Does NOT lower potassium
2. Shift Potassium into Cells
- IV insulin + glucose
- Nebulised salbutamol
- Sodium bicarbonate (if acidotic)
3. Remove Potassium from Body
- Loop diuretics
- Potassium binders
- Dialysis (definitive)
Dialysis Indications:
- Severe renal failure
- Refractory hyperkalaemia
- Life-threatening ECG changes
Important Points – Potassium
- Think arrhythmia first
- Always do ECG in hyperkalaemia
- Peaked T waves → early sign
- Wide QRS → pre-arrest
- Hypokalaemia → weakness, ileus, arrhythmia
- Correct magnesium if low
- K⁺ >6.5 or ECG changes → IV calcium immediately
Calcium Imbalance
Overview
Calcium is a vital extracellular cation essential for:
- Neuromuscular transmission
- Muscle contraction
- Blood coagulation
- Hormone secretion
- Enzyme activity
- Bone mineralization
Normal Values:
- Total serum calcium: 2.2–2.6 mmol/L (8.8–10.4 mg/dL)
- Ionized calcium: 1.1–1.3 mmol/L (physiologically active form)
Always interpret calcium in relation to serum albumin.
Corrected Calcium Formula
Corrected Calcium = Serum Calcium (mg/dL) + 0.8 × (4.0 − Serum Albumin (g/dL))
Calcium Homeostasis
Regulated by:
1. Parathyroid Hormone (PTH)
- ↑ Bone resorption
- ↑ Renal calcium reabsorption
- ↑ Phosphate excretion
- ↑ Vitamin D activation
2. Vitamin D (1,25-dihydroxyvitamin D)
- ↑ Intestinal calcium absorption
- ↑ Bone mineralization
Organs involved:
- Bone
- Kidney
- Gastrointestinal tract
Hypercalcaemia
Definition
Serum calcium > 2.6 mmol/L
Common Causes
1. Primary Hyperparathyroidism (Most common overall)
- Usually due to: Parathyroid adenoma
2. Malignancy (Most common inpatient cause)
- Mechanisms:
- PTHrP secretion (e.g., lung, renal cancers)
- Bone metastases
- Myeloma
Other Causes:
- Vitamin D excess
- Thiazide diuretics
- Granulomatous disease (e.g., Sarcoidosis)
- Hyperthyroidism
- Immobilization
- Addison’s disease
Clinical Features
“Stones, Bones, Groans and Psychic Moans”
- Renal stones
- Bone pain
- Abdominal pain, constipation
- Depression, confusion
- Polyuria & polydipsia
- Shortened QT interval (ECG)
Severe hypercalcaemia (>3.5 mmol/L) → Medical emergency
Investigation
- Repeat calcium
- PTH level (key investigation)
- High/inappropriately normal → Primary hyperparathyroidism
- Suppressed → Malignancy or other causes
- Urea & electrolytes
- Vitamin D
- Myeloma screen
- CXR (malignancy, sarcoidosis)
Management
Acute Severe Hypercalcaemia:
- IV 0.9% saline (first line)
- IV bisphosphonates (e.g., zoledronic acid)
- Calcitonin (short-term)
- Steroids (if vitamin D-mediated)
- Dialysis (if refractory)
Definitive Treatment:
- Parathyroidectomy (if primary hyperparathyroidism and symptomatic)
Indications for surgery:
- Symptomatic
- Ca >0.25 mmol/L above normal
- Osteoporosis
- Renal stones
- Age <50
Hypocalcaemia
Definition
Serum calcium < 2.2 mmol/L
Causes
1. Hypoparathyroidism
- Post-thyroidectomy (most common)
- Autoimmune
- Genetic
2. Vitamin D Deficiency
- Common in elderly and malnourished
3. Chronic Kidney Disease
- ↓ Vitamin D activation
- ↑ Phosphate retention
4. Acute Pancreatitis
5. Massive Blood Transfusion (citrate binding)
Clinical Features
Due to increased neuromuscular excitability:
- Perioral numbness
- Tetany
- Muscle cramps
- Seizures
- Laryngospasm
Classical Signs:
- Chvostek’s sign
- Trousseau’s sign
ECG: Prolonged QT interval
Investigation
- Serum calcium (corrected)
- PTH
- Vitamin D
- Magnesium (important!)
- Phosphate
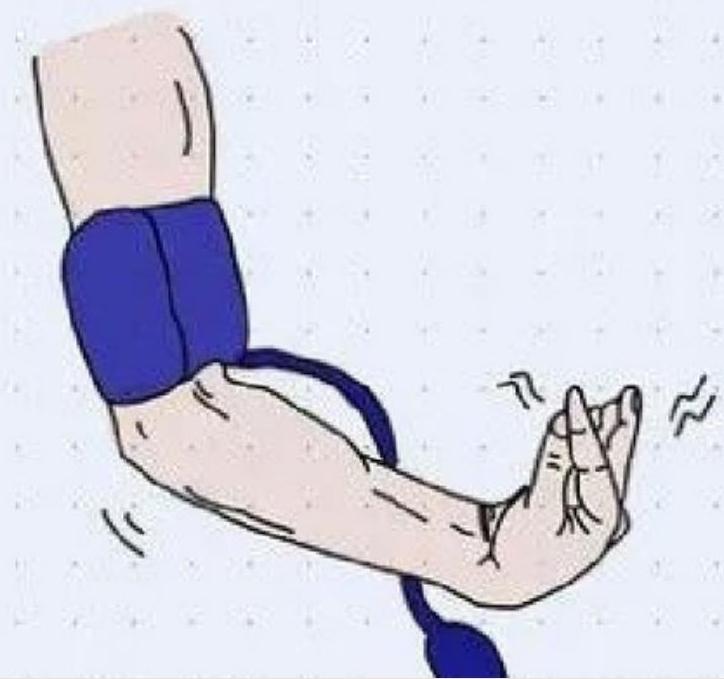
Trousseau’s Sign
How to test:
- Place blood pressure cuff around the arm
- Inflate the cuff for 1–4 mins
- If hands & fingers go into spasm in palmar flexion = positive
Chvostek’s Sign
How to test:
- Tap the face just below and in front of the ear
- If facial twitching occurs of one side of the mouth, nose & cheek = positive
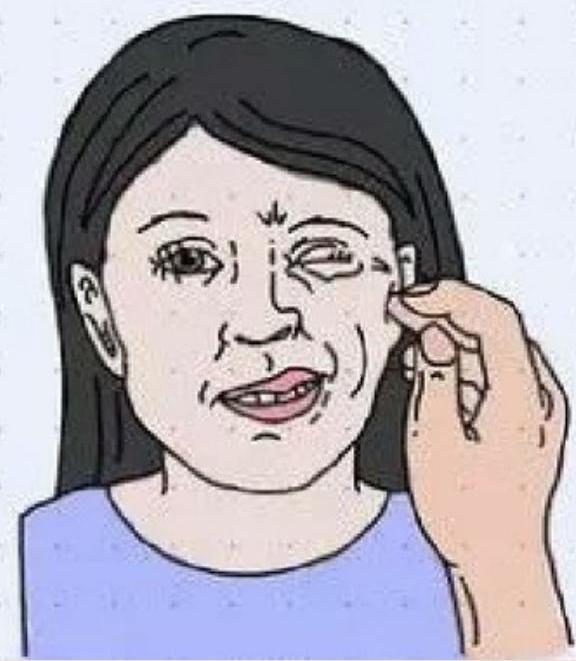
Management
Acute Symptomatic Hypocalcaemia:
- IV calcium gluconate (10%) slow infusion
- Cardiac monitoring
Chronic Management:
Depends on cause:
- Oral calcium supplements
- Vitamin D (cholecalciferol)
- Calcitriol in CKD
- Magnesium replacement if low
Secondary & Tertiary Hyperparathyroidism
Secondary Hyperparathyroidism
Due to chronic hypocalcaemia (usually CKD)
- High PTH
- Low/normal calcium
Tertiary Hyperparathyroidism
Autonomous PTH secretion after long-standing secondary hyperparathyroidism
- High PTH
- High calcium
Important Points – Calcium
- Correct calcium for albumin
- Hypocalcaemia → tetany, seizures, prolonged QT
- Hypercalcaemia → “bones, stones, groans, psychiatric overtones”
- Severe hypercalcaemia → dehydration + confusion
- First-line treatment (severe hypercalcaemia) → IV fluids
Sodium Imbalance
Physiology of Sodium Balance
Key Principles
- Sodium (Na⁺) is the major extracellular cation.
- Normal serum sodium: 135–145 mmol/L
- Determines plasma osmolality.
Plasma Osmolality Formula
Sodium reflects water balance, not total body sodium alone.
Regulation
- ADH (Vasopressin) – regulates water reabsorption
- Thirst mechanism
- Renin–angiotensin–aldosterone system (RAAS)
- Natriuretic peptides
Hyponatraemia (Na <135 mmol/L)
Definition
Serum sodium <135 mmol/L
| Severity | Range |
|---|---|
| Mild | 130–134 mmol/L |
| Moderate | 120–129 mmol/L |
| Severe | <120 mmol/L |
Stepwise Diagnostic Approach
Step 1: Confirm True Hyponatraemia
Check serum osmolality.
A. Hypotonic hyponatraemia (most common)
- Low sodium + low osmolality
B. Isotonic (Pseudohyponatraemia)
- Hyperlipidaemia
- Hyperproteinaemia
C. Hypertonic hyponatraemia
- Hyperglycaemia
- Mannitol
Step 2: Assess Volume Status
1. Hypovolaemic Hyponatraemia
Loss of sodium > water
Causes:
- Vomiting/diarrhoea
- Thiazide diuretics
- Addison’s disease
- Third spacing
Urine sodium:
- <20 mmol/L → extrarenal loss
- >20 mmol/L → renal loss
2. Euvolaemic Hyponatraemia
No oedema, no dehydration
Most common cause: SIADH (Syndrome of Inappropriate ADH)
Causes of SIADH:
- CNS disease
- Lung pathology (e.g., pneumonia)
- Drugs (SSRIs, carbamazepine)
- Malignancy (classically Small-cell lung carcinoma)
Diagnostic criteria:
- Low plasma osmolality
- Inappropriately concentrated urine
- Urine Na >30 mmol/L
- Normal adrenal and thyroid function
Other causes:
- Hypothyroidism
- Glucocorticoid deficiency
3. Hypervolaemic Hyponatraemia
Water retention > sodium retention
Seen in:
- Heart failure
- Cirrhosis
- Nephrotic syndrome
Mechanism: Effective arterial blood volume ↓ → ADH
Clinical Features of Hyponatraemia
Depends on:
- Severity
- Speed of onset
Acute (<48 hrs):
- Headache
- Confusion
- Seizures
- Coma
Due to cerebral oedema
Chronic:
- Often asymptomatic
Management of Hyponatraemia
Key Principle: Correct Slowly
Overcorrection → Osmotic Demyelination Syndrome (Central Pontine Myelinolysis): dysphagia, dysarthria, paralysis, encephalopathy and coma
Severe symptomatic hyponatraemia:
- 3% hypertonic saline
- Raise Na by 4–6 mmol/L initially
Max correction:
- <10 mmol/L in 24 hrs
- <18 mmol/L in 48 hrs
By volume status:
- Hypovolaemic → 0.9% saline
- SIADH:
- Fluid restriction (800–1000 mL/day)
- Treat cause
- Consider:
- Demeclocycline
- Tolvaptan (V2 antagonist)
- Hypervolaemic:
- Fluid restriction
- Loop diuretics
- Treat underlying condition
Hypernatraemia (Na >145 mmol/L)
Definition
Serum sodium >145 mmol/L
Always indicates water deficit relative to sodium
Causes
1. Water Loss (most common)
A. Renal Loss:
- Osmotic diuresis
- Diabetes insipidus
- Central DI: ↓ ADH production
- Nephrogenic DI: Kidney resistant to ADH
- Water deprivation test differentiates
B. Extrarenal Loss:
- Diarrhoea
- Burns
- Sweating
2. Sodium Gain:
- Hypertonic saline
- Sodium bicarbonate excess
Clinical Features
- Thirst
- Lethargy
- Irritability
- Seizures
- Coma
Due to cellular dehydration
Management of Hypernatraemia
Key Rule: Correct Slowly
Rapid correction → cerebral oedema
Calculate Water Deficit:
Water deficit = TBW × (current Na/140 − 1)
Where TBW:
- 0.6 × weight (men)
- 0.5 × weight (women)
Treatment:
- Free water orally if possible
- IV 5% dextrose
- 0.45% saline
Correct over 48–72 hours
Important Points – Sodium
- Symptoms depend on speed of change
- Always assess volume status
- Severe hyponatraemia → seizures, coma
- Correct slowly → prevent osmotic demyelination syndrome
- Hypernatraemia = usually water loss
- Correct hypernatraemia slowly → prevent cerebral oedema
- Confusion in elderly/on diuretics → check Na
Magnesium Imbalance
Overview
- Normal serum magnesium: 0.7–1.0 mmol/L
- 50–60% stored in bone
- Regulated mainly by:
- Kidneys (primary regulator)
- Gastrointestinal absorption
Magnesium is essential for:
- Neuromuscular stability
- ATP-dependent reactions
- Potassium and calcium homeostasis
Hypomagnesaemia (<0.7 mmol/L)
Causes
1. Reduced intake/absorption
- Malnutrition
- Chronic alcoholism
- Chronic diarrhoea
- Proton pump inhibitors (long-term)
2. Increased renal loss
- Diuretics (loop, thiazide)
- Uncontrolled diabetes
- Hypercalcaemia
- Aminoglycosides
Clinical Features
- Often associated with hypokalaemia and hypocalcaemia
- Neuromuscular hyperexcitability:
- Tremors
- Tetany
- Seizures
- Cardiac arrhythmias (e.g., torsades de pointes)
Management
- Correct underlying cause
- Oral magnesium if mild
- IV magnesium sulfate if severe or symptomatic
- Always correct magnesium in refractory hypokalaemia
Hypermagnesaemia (>1.0 mmol/L)
Causes
- Renal failure (most common)
- Excess magnesium intake (e.g., antacids, laxatives)
- Iatrogenic (e.g., obstetric therapy)
Clinical Features (dose-dependent)
- Nausea, flushing
- Loss of deep tendon reflexes
- Hypotension
- Bradycardia
- Respiratory depression
- Cardiac arrest (severe)
Management
- Stop magnesium intake
- IV calcium gluconate (cardiac stabilisation)
- IV fluids + loop diuretics
- Dialysis if severe (especially in renal failure)
Important Points – Magnesium
- Hypomagnesaemia causes refractory hypokalaemia
- Always check magnesium in unexplained arrhythmias
- Loss of reflexes = early sign of hypermagnesaemia
- Renal failure is the commonest cause of hypermagnesaemia
Chloride Imbalance
Overview
Chloride (Cl⁻) is the major extracellular anion.
Normal serum chloride: 98–106 mmol/L
It plays a key role in:
- Maintaining electroneutrality
- Acid-base balance
- Osmotic pressure
Clinically, chloride disorders are usually secondary to acid-base disturbances.
Hypochloraemia (Cl⁻ < 98 mmol/L)
Common Causes
- Vomiting / Nasogastric suction — Loss of HCl → metabolic alkalosis
- Diuretics (loop, thiazide)
- Chronic respiratory acidosis (renal compensation)
Clinical Features
Usually due to associated alkalosis:
- Weakness
- Tetany (if alkalosis severe)
- Volume depletion
Management
- Treat underlying cause
- IV normal saline (chloride replacement)
- Correct potassium if low
Hyperchloraemia (Cl⁻ > 106 mmol/L)
Common Causes
- Metabolic acidosis (normal anion gap) — Diarrhoea
- Renal tubular acidosis
- Excess IV normal saline (iatrogenic)
- Renal failure
Clinical Features
Due to metabolic acidosis:
- Kussmaul breathing
- Weakness
- Confusion
Management
- Treat underlying cause
- Adjust IV fluids
- Correct acidosis if severe
Important Points – Chloride
- Vomiting → hypochloraemic metabolic alkalosis
- Diarrhoea → hyperchloraemic metabolic acidosis
- Always interpret chloride with bicarbonate
- Chloride abnormalities reflect acid-base disorders rather than primary disease
Bicarbonate Imbalance
Overview
Bicarbonate (HCO₃⁻) is the major extracellular buffer.
Normal plasma bicarbonate: 22–28 mmol/L
It is regulated by:
- Kidneys (metabolic component)
- Lungs (via CO₂)
Changes in bicarbonate primarily reflect metabolic acid-base disorders.
Low Bicarbonate (Metabolic Acidosis)
Causes
A. High Anion Gap Metabolic Acidosis (HAGMA)
- Diabetic ketoacidosis
- Lactic acidosis
- Renal failure
- Toxins (e.g., salicylates)
B. Normal Anion Gap (Hyperchloraemic) Acidosis
- Diarrhoea
- Renal tubular acidosis
Clinical Features
- Kussmaul respiration
- Confusion
- Hypotension (severe cases)
Management
- Treat underlying cause
- IV fluids
- Insulin (DKA)
- Bicarbonate only if severe acidosis (pH < 7.1)
High Bicarbonate (Metabolic Alkalosis)
Causes
- Vomiting (loss of gastric acid)
- Diuretics
- Excess alkali intake
- Hyperaldosteronism
Clinical Features
- Weakness
- Tetany (due to ↓ ionized calcium)
- Arrhythmias (often with hypokalaemia)
Management
- Correct volume depletion (IV saline)
- Replace potassium
- Treat underlying cause
Important Points – Bicarbonate
- Low HCO₃⁻ = metabolic acidosis
- High HCO₃⁻ = metabolic alkalosis
- Always calculate anion gap
- Interpret alongside pH and PaCO₂ (ABG)
- Vomiting → metabolic alkalosis
- Diarrhoea → metabolic acidosis
Phosphate Imbalance
Overview
- Normal serum phosphate: 0.8–1.5 mmol/L (2.5–4.5 mg/dL)
- 85% stored in bone (hydroxyapatite)
- Regulated by:
- Kidney (primary regulator)
- Parathyroid hormone (PTH)
- Vitamin D
- FGF-23
Phosphate balance depends on intestinal absorption, renal excretion, and bone exchange.
Hypophosphataemia (<0.8 mmol/L)
Causes
1. Reduced intake/absorption
- Malnutrition
- Alcoholism
- Vitamin D deficiency
- Antacids (phosphate binding)
2. Redistribution (intracellular shift)
- Refeeding syndrome
- Insulin therapy
- Respiratory alkalosis
3. Increased renal loss
- Hyperparathyroidism
- Fanconi syndrome
- Diuretics
Clinical Features
- Often asymptomatic (mild)
- Muscle weakness
- Rhabdomyolysis
- Respiratory failure
- Haemolysis
- Confusion, seizures (severe)
Management
- Treat cause
- Oral phosphate if mild
- IV phosphate if severe/symptomatic (monitor calcium closely)
Hyperphosphataemia (>1.5 mmol/L)
Causes
1. Reduced renal excretion (most common)
- Chronic kidney disease (CKD)
2. Increased load
- Tumour lysis syndrome
- Rhabdomyolysis
- Massive haemolysis
3. Endocrine causes
- Hypoparathyroidism
Clinical Features
- Usually asymptomatic
- Hypocalcaemia symptoms (tetany)
- Soft tissue and vascular calcification (chronic)
Management
- Treat underlying cause
- Dietary phosphate restriction
- Phosphate binders (e.g., calcium carbonate, sevelamer)
- Dialysis if severe (especially in CKD)
Important Points – Phosphate
- CKD = hyperphosphataemia
- Refeeding syndrome = hypophosphataemia
- Phosphate abnormalities often alter calcium levels inversely
- Severe hypophosphataemia can cause respiratory failure in ICU patients