Visceral Leishmaniasis
Dr. Eatimad Mahgoub Osheik
Learning Objectives
By the end of this lecture, you should be able to:
- Describe the epidemiology and life cycle of Leishmania
- Classify different types of leishmaniasis
- Understand pathogenesis of visceral leishmaniasis
- Recognize clinical features and complications
- Discuss diagnosis, treatment, and prevention
Introduction
Leishmaniasis is a protozoal disease with the following characteristics:
- Caused by Leishmania species
- Transmitted by the bite of female sandfly
- An important neglected tropical disease
- Affects skin, mucosa, or internal organs
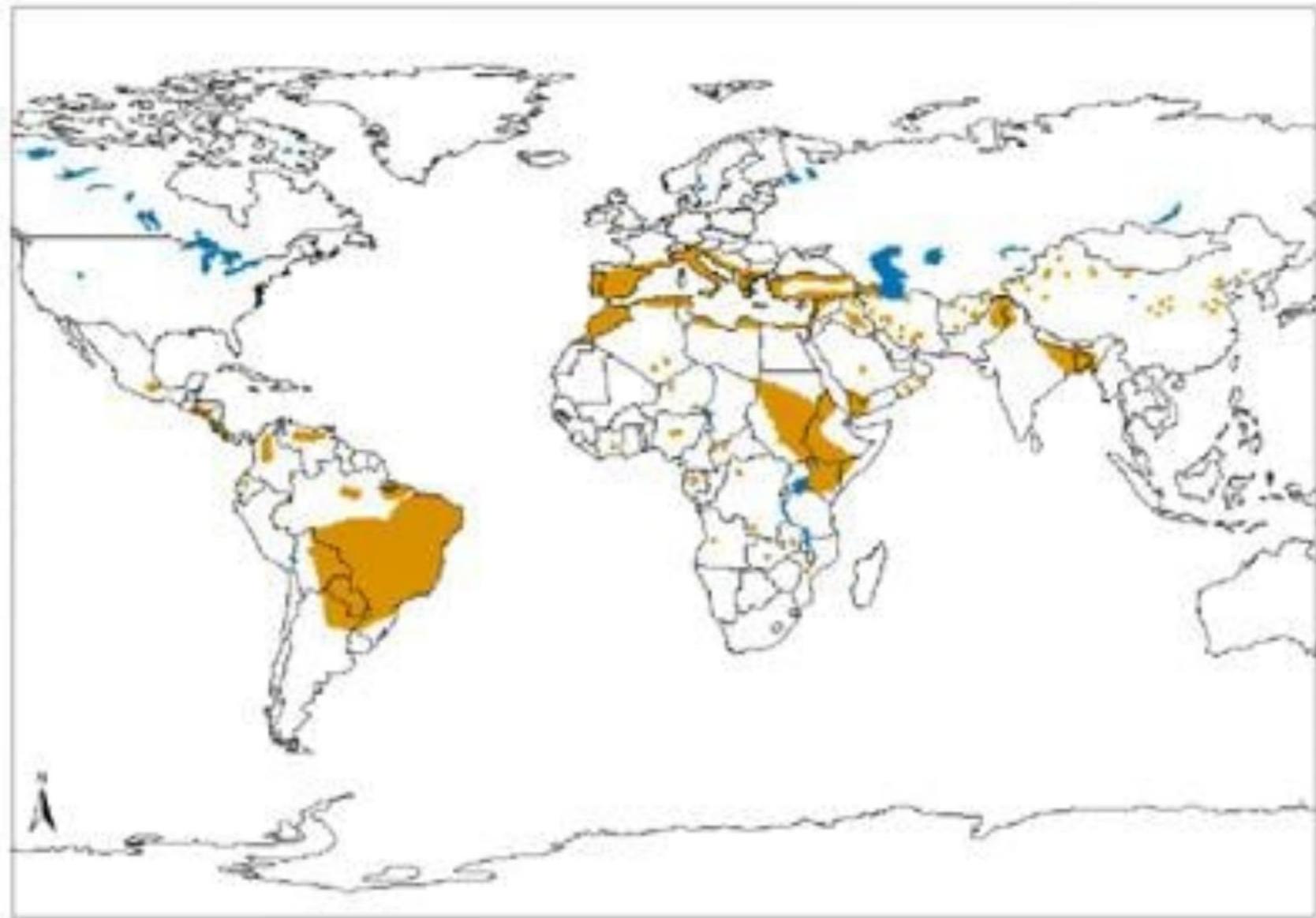
Global Epidemiology
Geographic Distribution:
- Endemic in tropical and subtropical regions
- Found in Asia, Africa, South America, and Mediterranean regions
- 90% of Visceral Leishmaniasis cases occur in:
- India
- Bangladesh
- Sudan
- Brazil
- Ethiopia
Disease Burden
- Prevalence: Around 12 million people infected globally
- Incidence: ~0.7–1.2 million Cutaneous Leishmaniasis cases and 0.2–0.4 million Visceral Leishmaniasis cases annually
- Mortality: VL is fatal if untreated (>95% of cases)
Risk Factors
Socioeconomic:
- Poverty
- Malnutrition
- Poor housing
- Lack of resources
Environmental:
- Rainforests
- Peri-urban areas
- Proximity to animal reservoirs (rodents)
Immune Status:
- Compromised immunity
- HIV co-infection (especially significant)
Human Behaviour:
- Population movement
- Lack of awareness
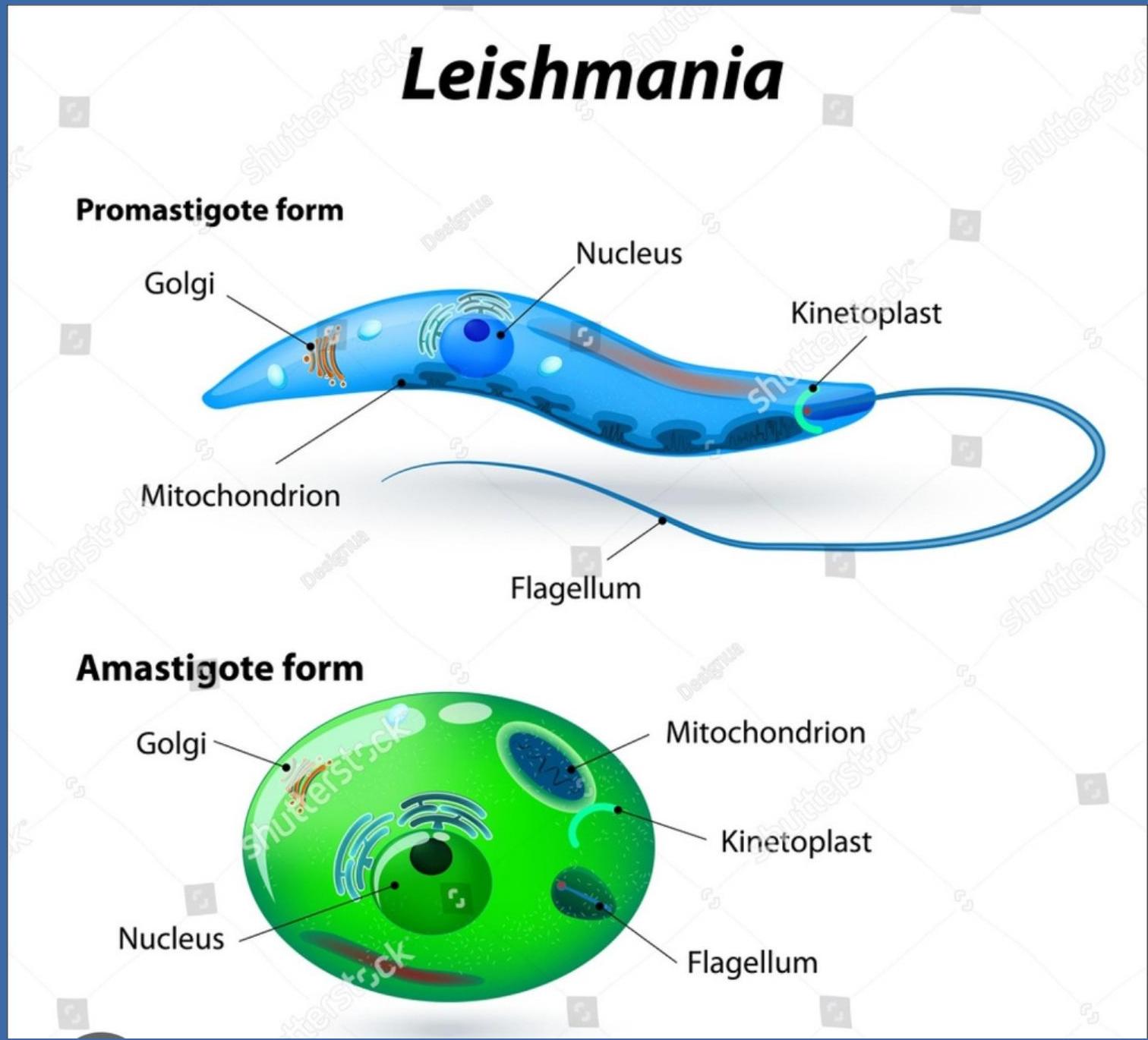
Causative Organism
Leishmania is an obligate intracellular protozoa that belongs to the Kinetoplastida class.
Old World Species (Africa, Asia, Europe)
- L. tropica, L. major, L. aethiopica → Cutaneous leishmaniasis
- L. donovani, L. infantum → Visceral leishmaniasis
New World Species (Americas)
- L. mexicana, L. amazonensis → Cutaneous / diffuse cutaneous leishmaniasis
- L. braziliensis → Mucocutaneous leishmaniasis
- L. chagasi (= L. infantum) → Visceral leishmaniasis
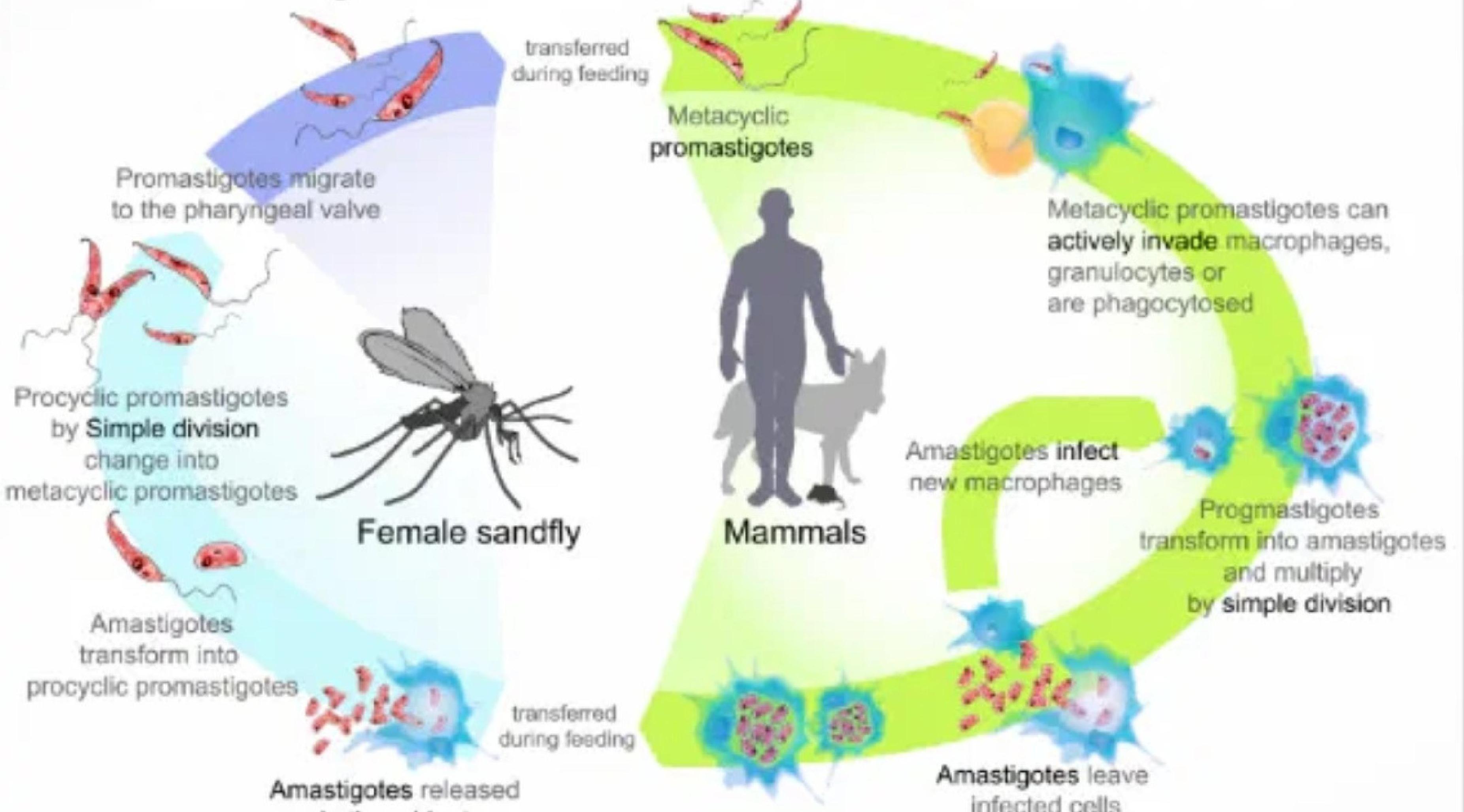
Life Cycle of Leishmania Parasite
Overview: The Leishmania parasite’s life cycle involves two hosts: a sandfly vector and a mammalian host. The parasite alternates between flagellated promastigotes (in the fly) and non-flagellated amastigotes (in the mammal).
Vector
Female Sandfly characteristics:
- Phlebotomus (Old World)
- Lutzomyia (New World)
- Small, silent, night-biting insect
- Breeds in:
- Cracks in walls
- Cattle sheds
- Organic waste

Leishmania Parasite Reservoir
Natural reservoirs include:
- Dogs
- Rodents
- Humans
- Wild animals (e.g., Foxes, Jackals)
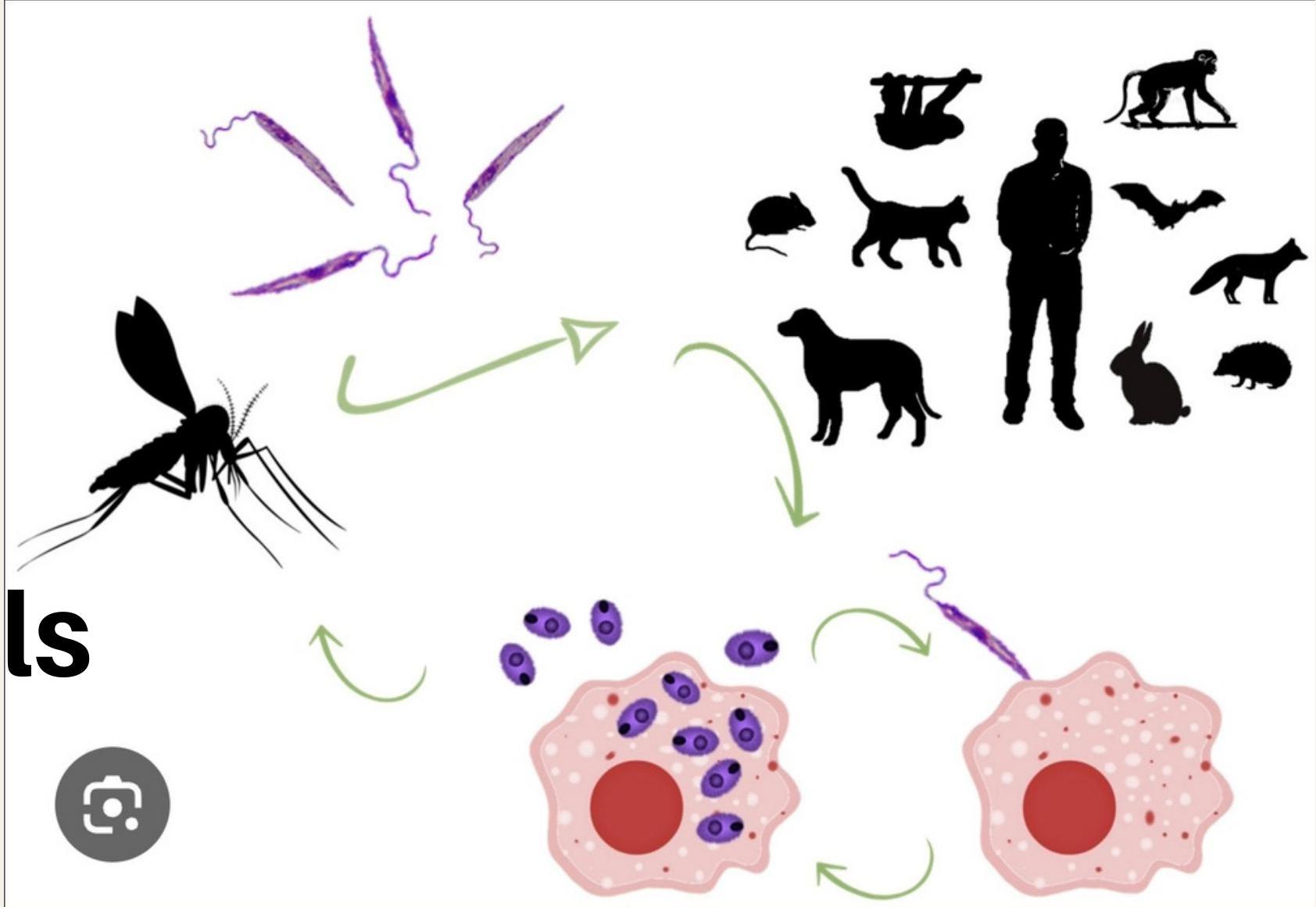
Life Cycle Steps
- Sandfly bites infected human → ingests amastigotes
- In sandfly gut → transform into promastigotes
- Promastigotes multiply and migrate to mouth
- Sandfly bites human → injects promastigotes
- Promastigotes enter macrophages → become amastigotes
- Multiply intracellularly → cell rupture → systemic spread
Target Organs
The parasite primarily targets:
- Spleen
- Liver
- Bone marrow
- Lymph nodes
Clinical Classification of Leishmaniasis
Based on Clinical Manifestations
- Visceral Leishmaniasis (VL)
- Cutaneous Leishmaniasis (CL)
- Mucocutaneous Leishmaniasis (MCL)
- Post-Kala-azar Dermal Leishmaniasis (PKDL)
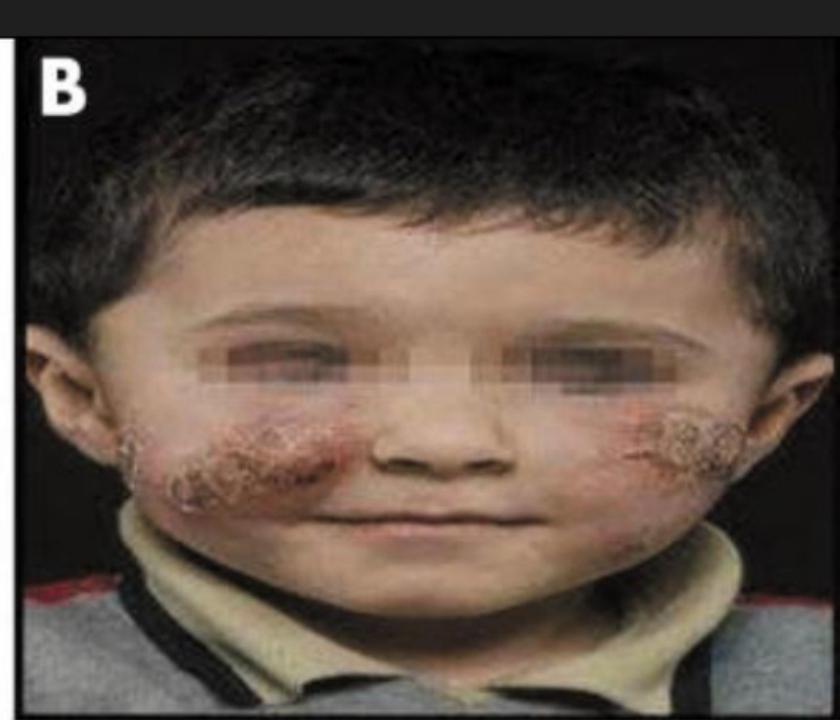
Cutaneous Leishmaniasis
Causative Organisms:
- L. tropica
- L. major
Clinical Features:
- Localized to skin
- Lesion progression: Papule → nodule → ulcer
- Usually painless
- Heals with scarring
- Known as “Oriental sore”
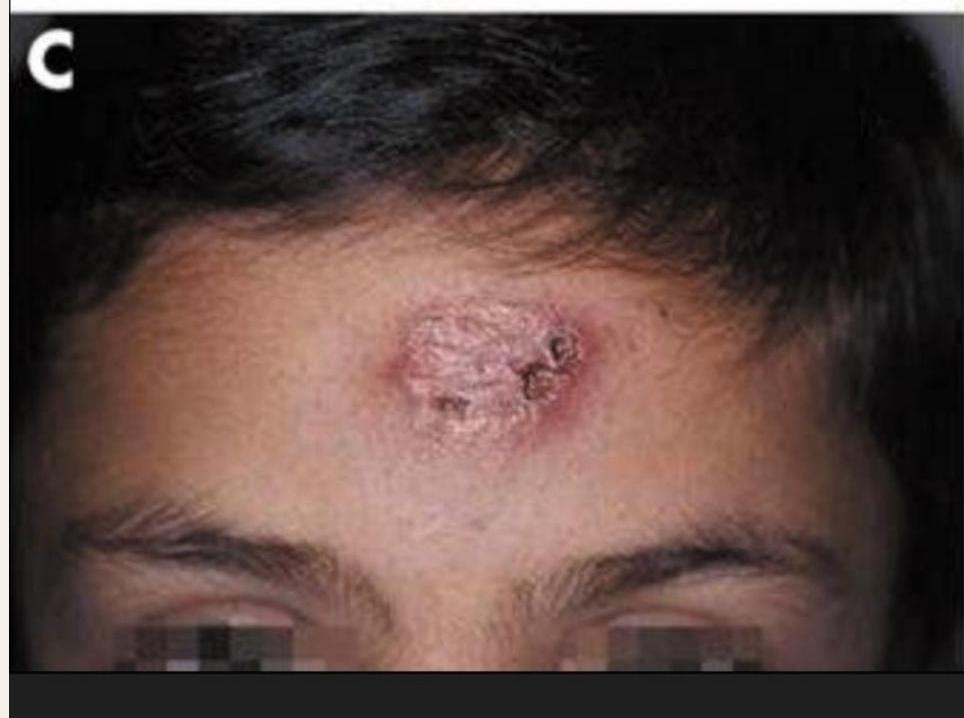
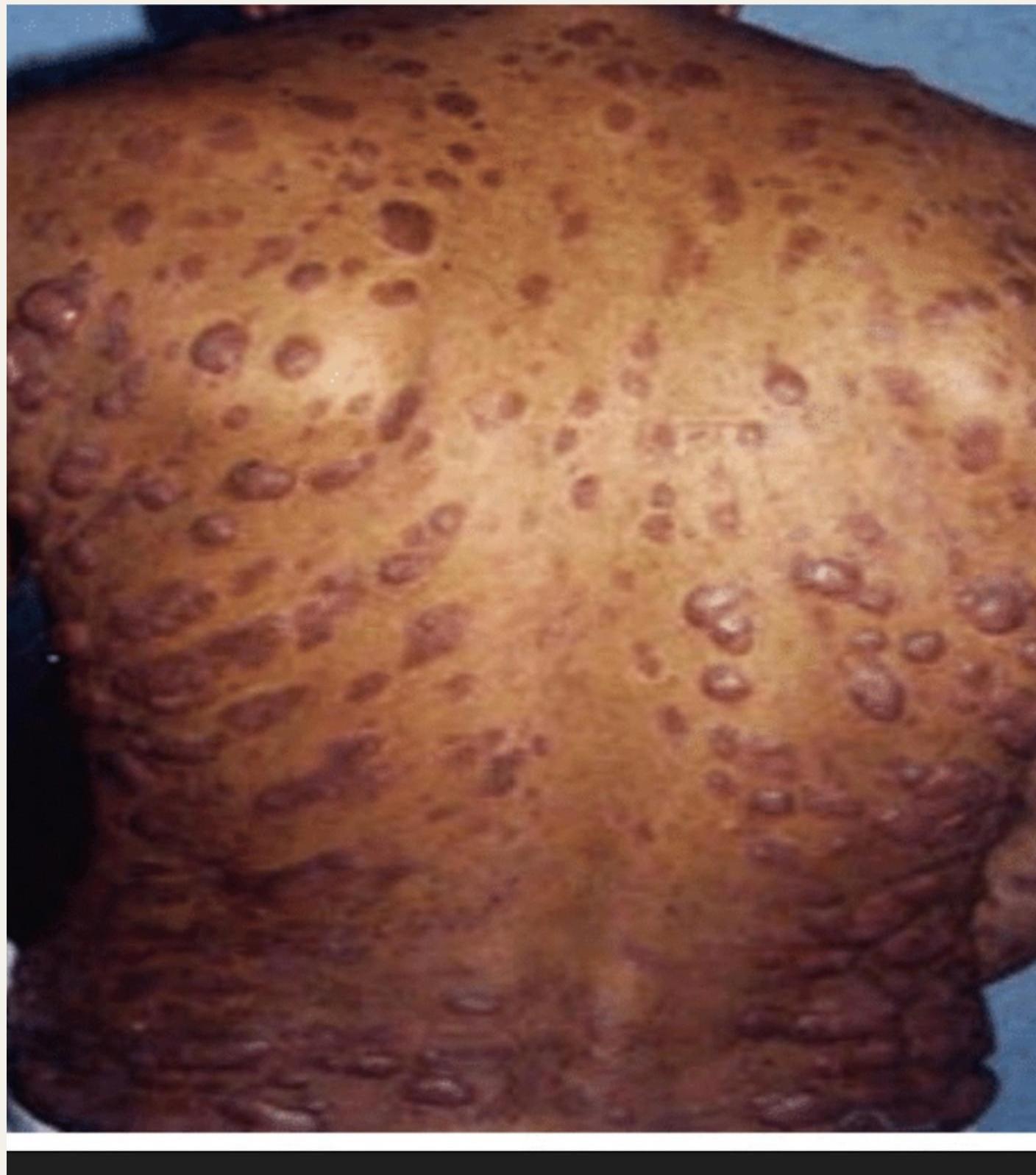
Diffuse Cutaneous Leishmaniasis
Characteristics:
- Rare form
- Associated with poor cell-mediated immunity
- Multiple nodular lesions
- Resembles lepromatous leprosy
- Difficult to treat
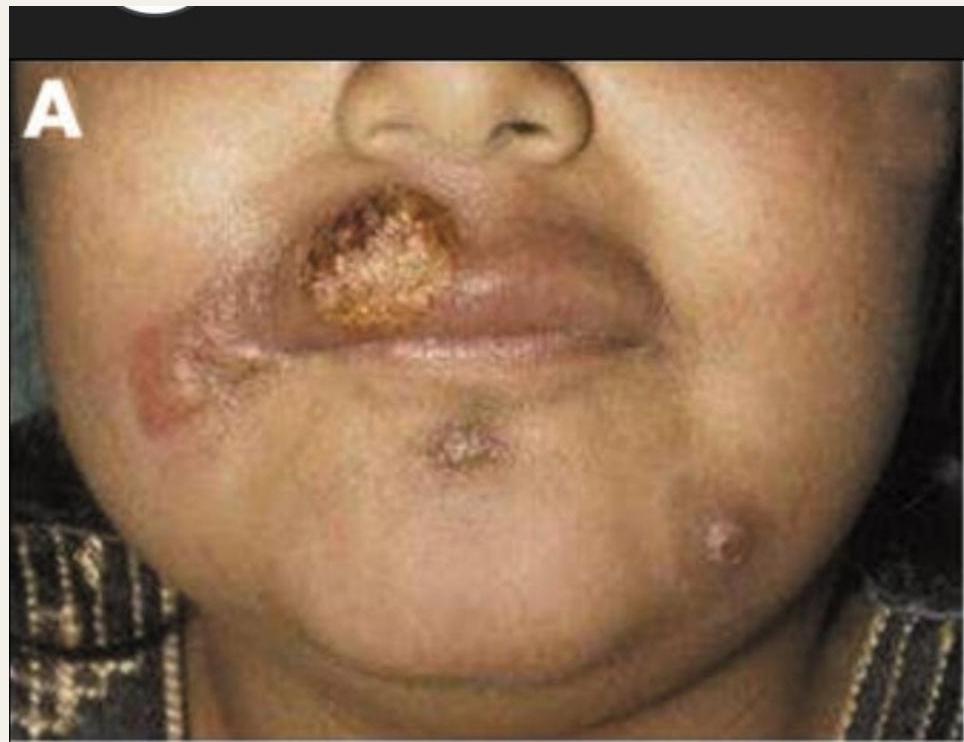
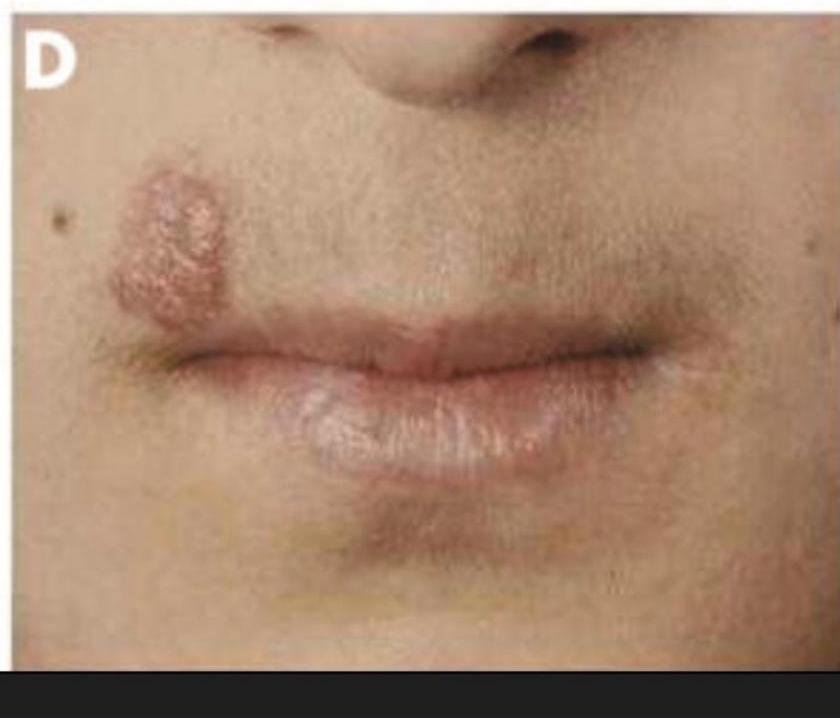
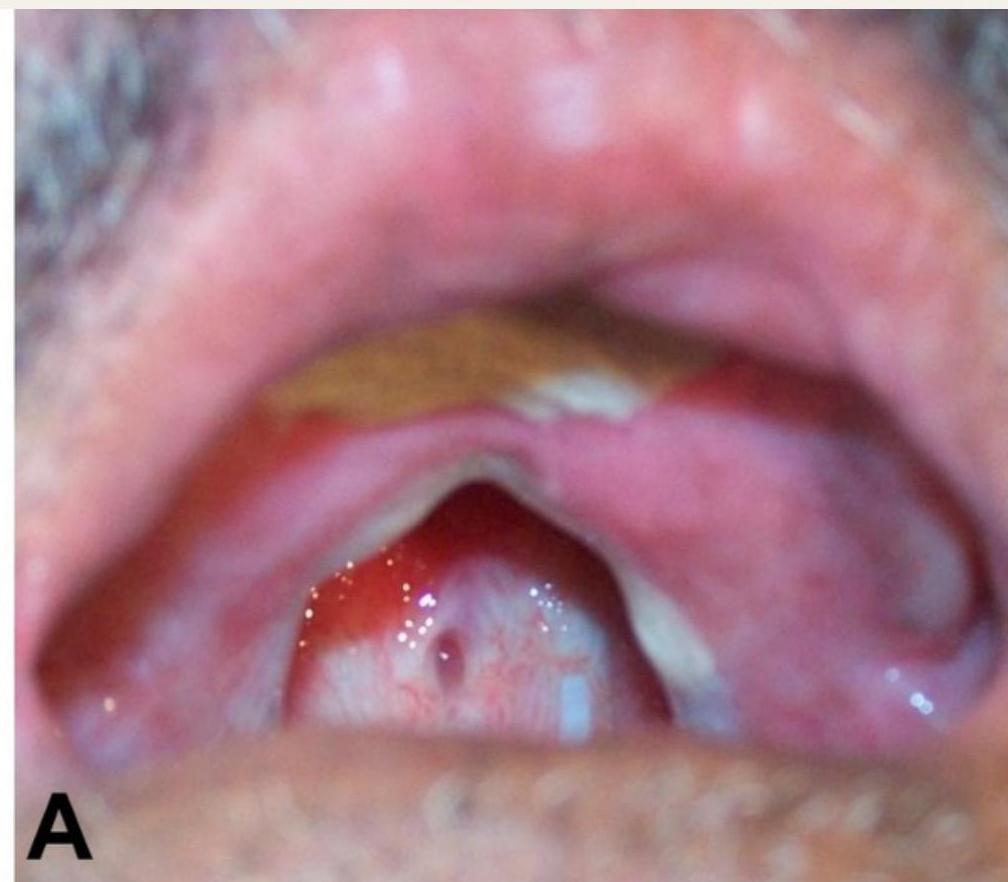
Mucocutaneous Leishmaniasis
Causative Organism:
- Leishmania braziliensis
Affected Structures:
- Nasal septum
- Oral cavity
- Pharynx
Consequences:
- Severe tissue destruction
- Facial disfigurement
- Common in South America
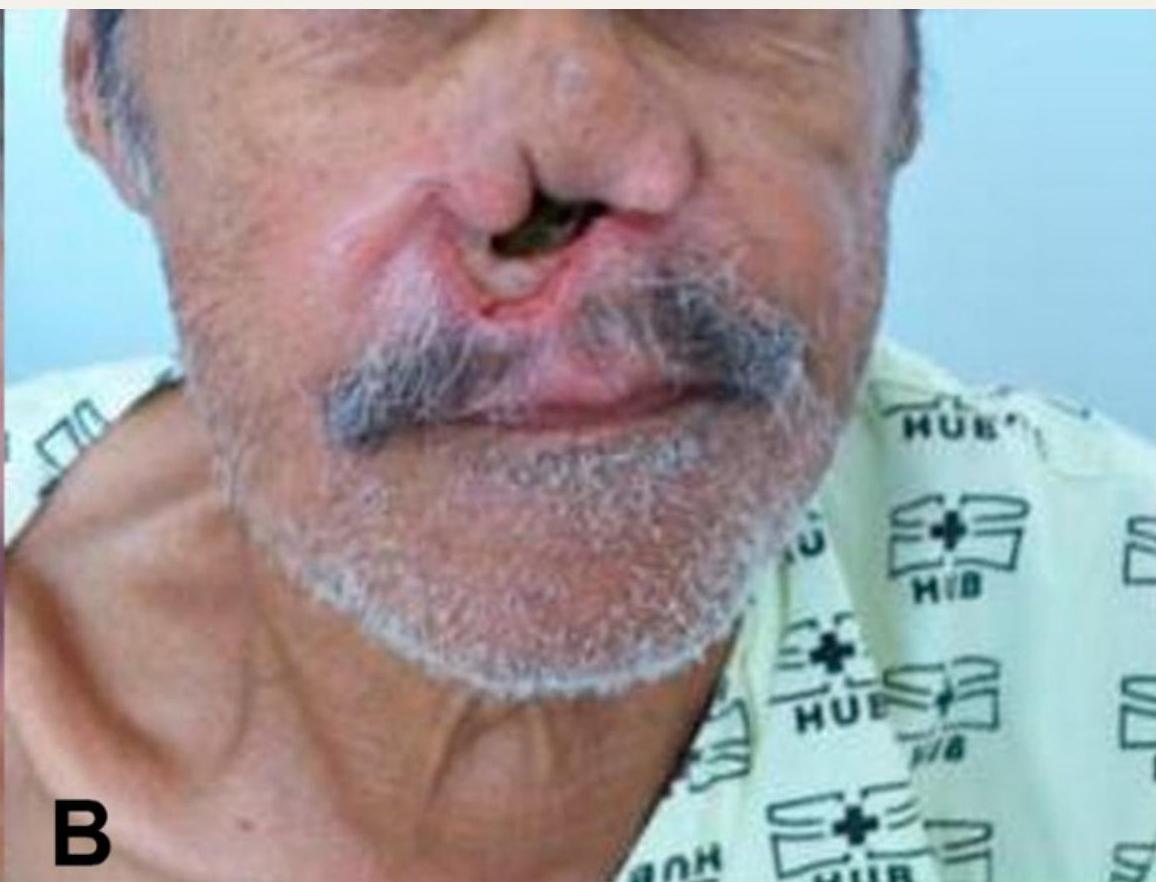
Post-Kala-azar Dermal Leishmaniasis (PKDL)
Timing:
- Occurs after treatment of VL
- Develops months to years post-treatment
Clinical Manifestations:
- Hypopigmented macules
- Papules
- Nodules
- No systemic symptoms
Epidemiological Significance:
- Serves as reservoir of infection
- Important in disease transmission
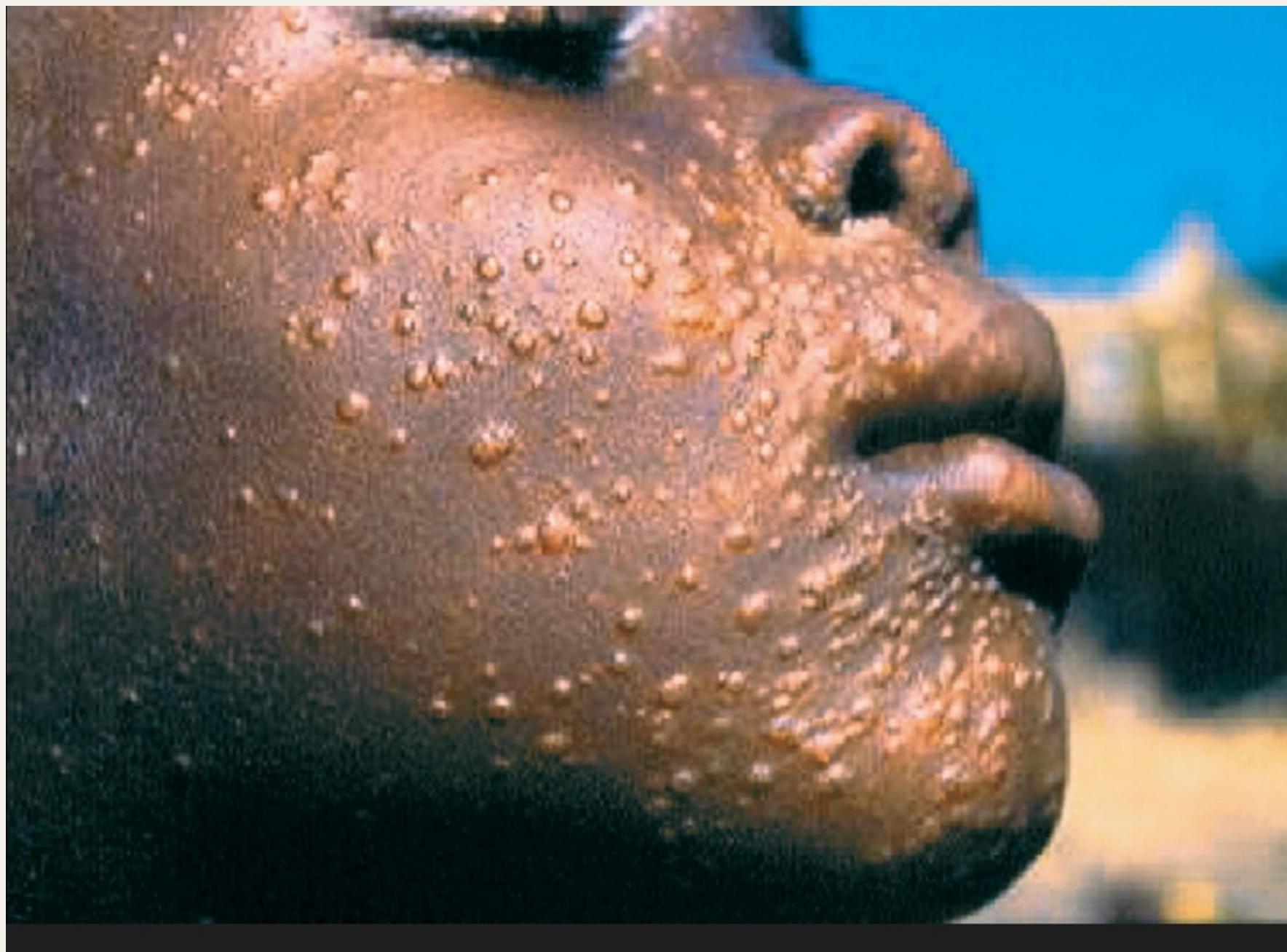
Visceral Leishmaniasis (VL) - “Kala-azar” (The Black Fever)
Overview:
- Severe, potentially fatal disease
- Caused by Leishmania donovani and Leishmania infantum (chagasi)
- Targets internal organs (spleen, liver, and bone marrow)
- High mortality if untreated
Pathogenesis of Visceral Leishmaniasis (Kala-azar)
Incubation Period:
- 2–6 months, but can extend up to 2 years
Mechanism of Spread:
- Amastigotes multiply within macrophages
- Spread via blood and lymphatics
- Parasites primarily invade the reticuloendothelial system (spleen, liver, bone marrow, and lymph nodes)
Systemic Manifestations:
- Splenomegaly
- Hepatomegaly
- Lymphadenopathy
- Anemia
- Leukopenia
- Thrombocytopenia
Immune Consequences:
- Progressive immune suppression occurs due to macrophage dysfunction
Effects of Visceral Leishmaniasis on the Immune System
Cellular Immunity Suppression:
- The parasite infects macrophages and suppresses protective cell-mediated (Th1) immunity
- Leads to reduced IFN-γ and IL-12 production
Immunosuppressive Response:
- Increased immunosuppressive cytokines (IL-10) which allow parasite survival
- Immune response shifts toward ineffective humoral (Th2) response - causing polyclonal B-cell activation and hypergammaglobulinemia
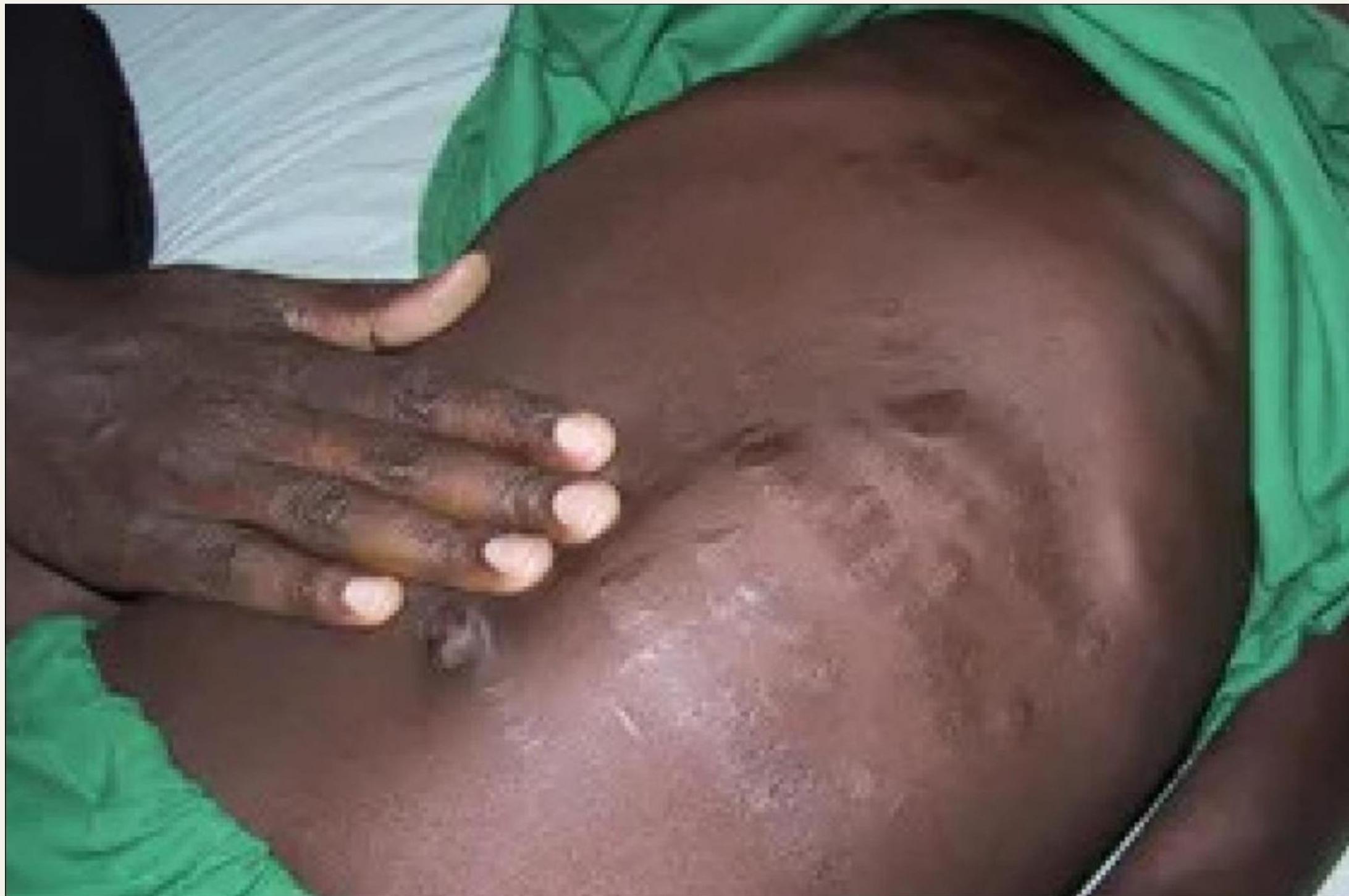
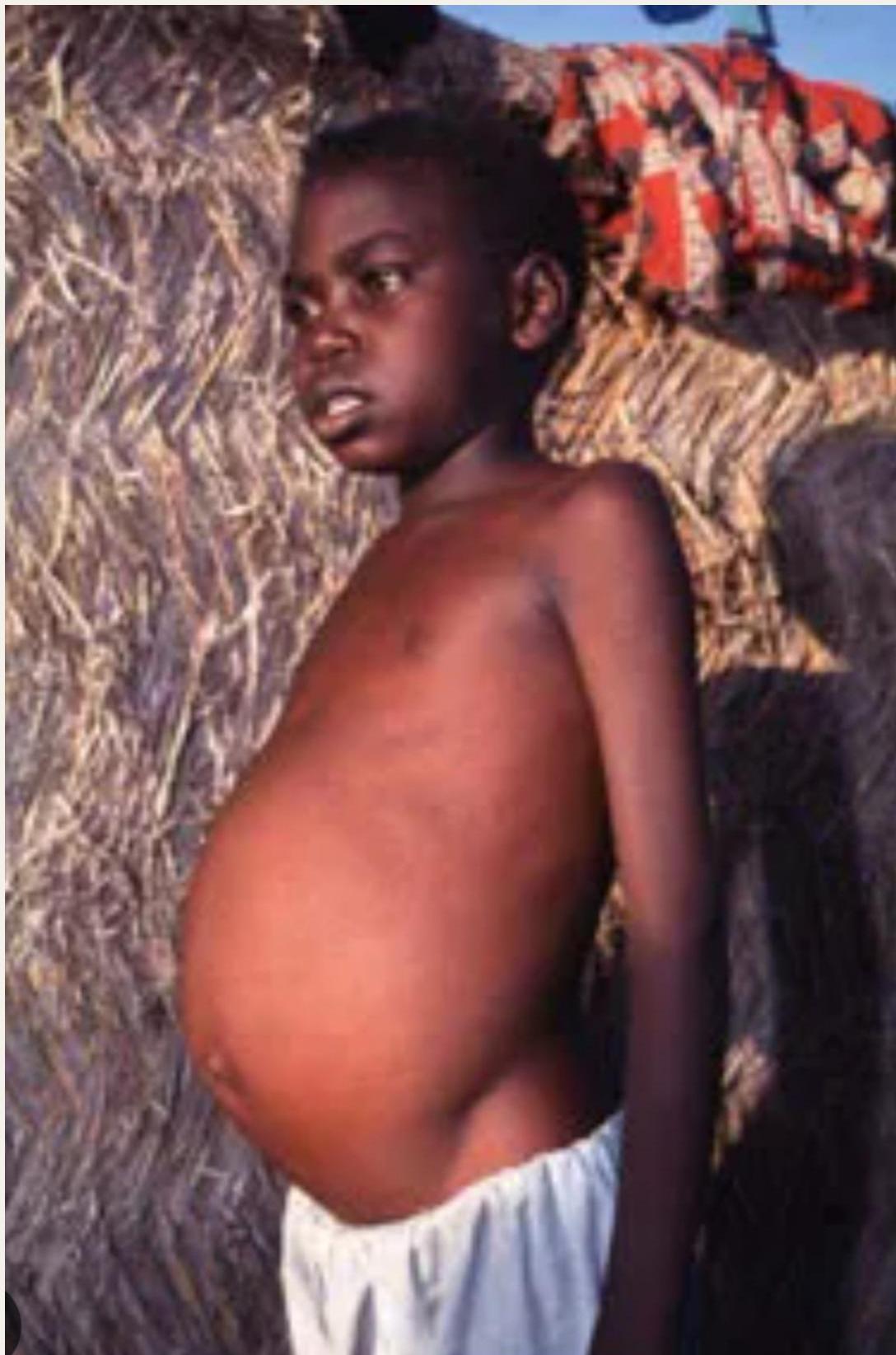
Clinical Presentation of Visceral Leishmaniasis
Fever:
- Irregular, prolonged bouts
Constitutional Symptoms:
- Progressive and severe weight loss
- General weakness
- Malaise
- Anorexia
- Fatigue
Physical Examination Findings:
- Hepatosplenomegaly: Enlarged, non-tender spleen and liver causing abdominal swelling
Laboratory Abnormalities:
- Pancytopenia: Anemia, leucopenia, thrombocytopenia
Dermatologic Manifestations:
- Skin darkening or spots
Hemorrhagic Manifestations:
- Bleeding: Petechiae or bruising
Diagnostic Studies

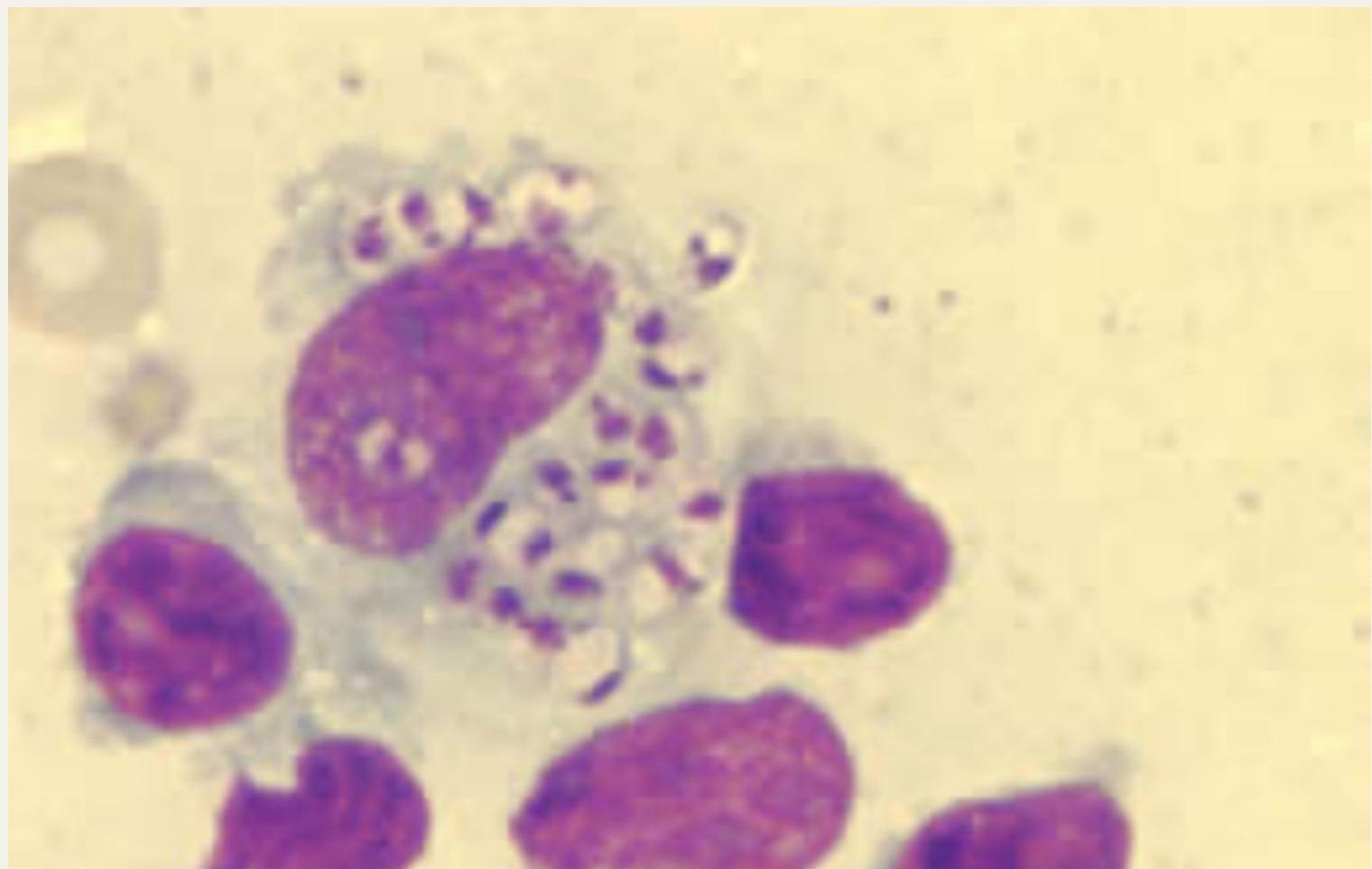
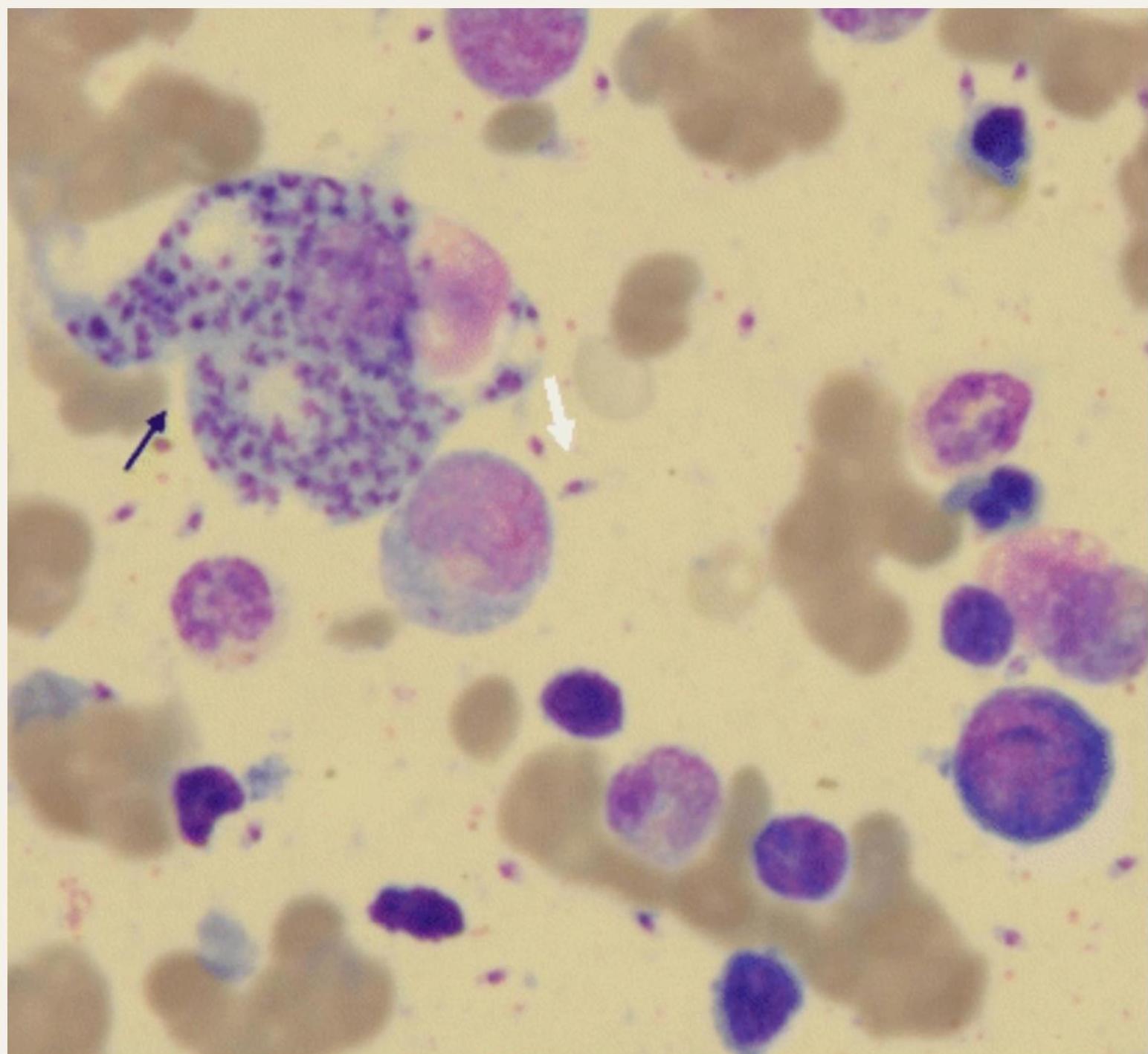
1. Parasitological Diagnosis (Gold Standard)
Detection Method: Detects Leishmania amastigotes (Leishman Donovan LD bodies)
Splenic Aspirate Microscopy:
- Sensitivity: ~95%
- Risk: Bleeding (needs expertise)
Bone Marrow Aspirate:
- Sensitivity: 60–85%
- Advantages: Safer, commonly used
Lymph Node Aspirate:
- Lower sensitivity
Culture:
- Confirms species identity
- Slow process
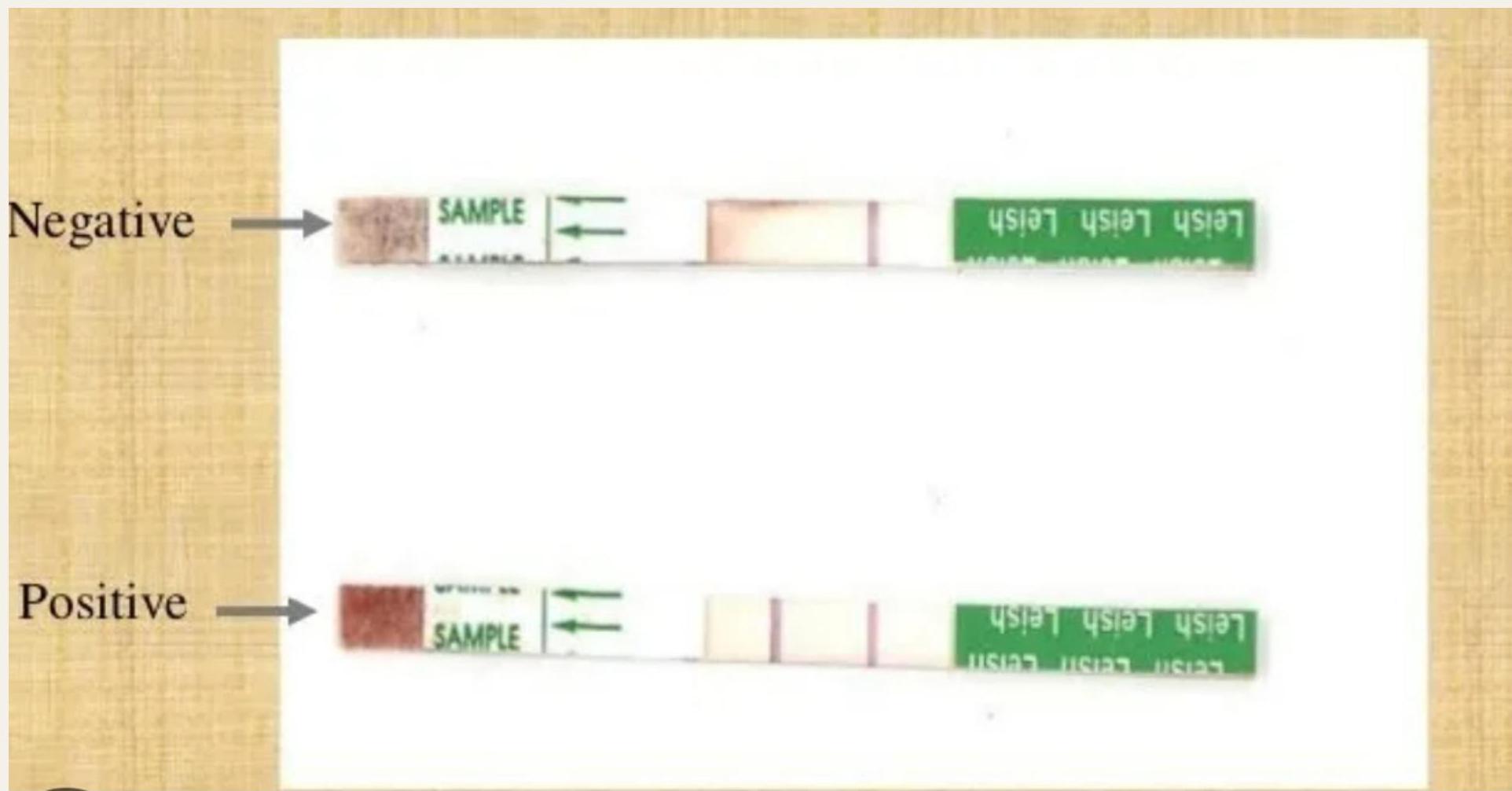
2. Serological Tests (Antibody Detection)
Useful for screening and diagnosis in endemic areas
rK39 Immunochromatographic Test (ICT):
- Rapid, field-friendly
- High sensitivity & specificity
Direct Agglutination Test (DAT):
- Highly sensitive
- Quantitative
ELISA:
- Sensitive
- Lab-based
IFA (Indirect Fluorescent Antibody Test):
- Specialized laboratory test
Limitations of Serological Tests:
- Cannot distinguish past vs. active infection
- Lower sensitivity in HIV co-infection
- Antibodies persist after cure
3. Antigen Detection
KAtex (Urine Latex Agglutination Test):
- Detects leishmanial antigen in urine
- Useful for treatment monitoring
- Lower sensitivity than rK39
4. Molecular Tests
PCR (Polymerase Chain Reaction):
- Performed on blood, bone marrow, splenic aspirate
- Very high sensitivity & specificity
- Useful in HIV-VL, relapse, and low parasite load scenarios
- Limitations: Expensive, limited availability
5. Skin Test
Montenegro (Leishmanin) Skin Test:
- Negative in active VL
- Becomes positive after cure
- Not useful for diagnosis of active disease
6. Supportive Laboratory Findings (Non-specific)
- Pancytopenia (anemia, leukopenia, thrombocytopenia)
- Hypergammaglobulinemia
- Elevated ESR (Erythrocyte Sedimentation Rate)
- Hypoalbuminemia

Management
Supportive Care
- Adequate nutrition
- Correction of anemia
- Treatment of secondary infections
- Antipyretics for fever
- Management of bleeding and thrombocytopenia
Treatment
WHO-Approved Medications for Visceral Leishmaniasis
Several medicines have been approved by the World Health Organization (WHO) to treat VL cases:
- Pentavalent antimony
- Amphotericin B deoxycholate
- Lipid formulation of amphotericin B (e.g., liposomal amphotericin B)
- Paromomycin
- Miltefosine
Note: Pentavalent antimony has been used for several decades and remains effective against VL.
Drugs Registered in Saudi Arabia:
- Pentavalent antimonials (sodium stibogluconate & meglumine antimoniate)
- Ambisome (liposomal amphotericin B)
Pentavalent Antimonials
Formulations:
- Sodium stibogluconate solution: 100 mg/ml
- Meglumine antimoniate solution: 81 mg/ml
Dosage:
- 20 mg/kg/day × 30 days
- Route: Intramuscular or intravenous infusion
Side Effects:
- Cardiotoxicity
- Fatal arrhythmia
Contraindications:
- Elderly patients
- Significant heart, liver, or kidney disease
- Pregnancy
Ambisome (Liposomal Amphotericin B)
Advantages:
- Similar efficacy to amphotericin B
- Significantly less toxic than standard formulation
Side Effects:
- Transient nephrotoxicity
- Thrombocytopenia
- Hypokalemia
Dosage:
- 3 mg/kg/day
- Route: Intravenous
Clinical Use:
- Recommended for resistant cases
- Use when pentavalent antimony’s are contraindicated
Complications of Visceral Leishmaniasis (Kala-azar)
- Overwhelming bacterial infections (most common cause of death)
- Severe bleeding
- Extreme weight loss
- Anemia
- Hepatosplenomegaly leading to organ failure
- Disfiguring skin issues (PKDL)
- Disease relapse
Prevention & Control
Individual Level
- Early diagnosis and treatment
- Use of bed nets
- Personal protection
Vector Control
- Indoor residual spraying (DDT / pyrethroids)
- Environmental sanitation
National Programs
- Kala-azar Elimination Programme
- Target: <1 case per 10,000 population




Vaccination Against Leishmaniasis
Current Status:
- No licensed vaccine available for human leishmaniasis
- Several candidates are in various stages of development
- Some showing promising results in animal models and early human trials
- DNA vaccines (e.g., LEISHDNAVAX) showing potential
Case Scenario
Clinical Presentation
A 32-year-old male farmer from Sudan presents to the outpatient department with the following complaints:
Chief Complaints:
- Prolonged fever for the past 8 weeks
- Fever is intermittent, associated with chills and night sweats
- Progressive weakness
- Significant weight loss
- Loss of appetite
- Increasing abdominal fullness
Physical Examination (O/E)
General Appearance:
- Looks ill, pale, and emaciated
- Temperature: 38.5°C
- Pulse: 98 beats/min
- Blood Pressure: 110/70 mmHg
Abdominal Examination:
- Spleen: Palpable 8 cm below the left costal margin
- Moderate hepatomegaly
Systemic Examination:
- Generalized lymphadenopathy
- Hyperpigmentation of the skin
Laboratory Investigations
Hematologic Parameters:
- Hemoglobin: 8.5 g/dL
- Total leukocyte count: 2,500/mm³
- Platelet count: 90,000/mm³
Serological & Parasitological Testing:
- rK39 rapid test: Positive
- Bone marrow aspiration: Presence of LD bodies
Biochemical Parameters:
- Liver function tests: Mildly elevated
Diagnosis: Visceral Leishmaniasis (Kala-azar) caused by Leishmania donovani
Diagnostic Reasoning
This patient presents the classic triad of VL:
| Clinical Feature | Patient Finding | Pathophysiological Basis |
|---|---|---|
| Demographics | 32-year-old male farmer from Sudan | Sudan is among the 5 countries accounting for 90% of global VL cases (endemic region) |
| Prolonged fever | 8 weeks, intermittent with chills | Immune dysregulation with macrophage dysfunction; persistent parasitemia |
| Hepatosplenomegaly | Massive splenomegaly (8 cm), moderate hepatomegaly | Parasite invasion of reticuloendothelial system (spleen, liver, bone marrow) |
| Pancytopenia | Hb 8.5 g/dL, WBC 2,500/μL, Plt 90,000/μL | Bone marrow suppression + hypersplenism + peripheral destruction |
| Constitutional symptoms | Severe weight loss, anorexia, weakness | Chronic inflammation, cytokine storm (IL-10 elevation), malnutrition |
| Skin changes | Hyperpigmentation (“Black fever”) | Accumulation of melanin; characteristic of kala-azar |
| Lymphadenopathy | Generalized | Reticuloendothelial proliferation |
| Confirmatory tests | rK39 Positive (+), LD bodies in bone marrow | rK39 ICT is highly sensitive/specific for VL; visualization of amastigotes (LD bodies) in bone marrow aspirate is the gold standard (60–85% sensitive) |
Differential Diagnoses (Ruled Out)
- Malaria: No mention of cyclic fever patterns or peripheral smear findings; rK39 is specific for leishmaniasis
- Typhoid: Prolonged fever but would present with relative bradycardia, rose spots, and negative rK39
- Tuberculosis (Disseminated): Would show granulomas, not LD bodies, in bone marrow; chest imaging typically abnormal
- Lymphoma/Hematologic malignancy: Would present with lymphadenopathy but bone marrow would show malignant cells, not intracellular LD bodies; rK39 would be negative
- Schistosomiasis: Can cause hepatosplenomegaly and abdominal fullness, but typically causes eosinophilia (not pancytopenia) and negative serology for Leishmania
Management Plan
1. Supportive Care (Immediate)
- Transfusion: Packed red blood cells if Hb <8 g/dL with symptoms (weakness, pallor)
- Nutritional rehabilitation: High-calorie diet; address severe malnutrition
- Infection surveillance: Broad-spectrum antibiotics prophylactically given high risk of overwhelming bacterial infections (most common cause of death in VL)
- Platelet monitoring: Risk of hemorrhage with Plt 90,000/μL; avoid IM injections until platelet recovery
2. Specific Anti-Leishmanial Therapy
First-line options based on Saudi Arabian availability and WHO guidelines:
Option A (Preferred for this case): Liposomal Amphotericin B (Ambisome)
- Dose: 3 mg/kg/day IV infusion
- Advantages: Less nephrotoxic than conventional amphotericin B; safer than antimonials for prolonged therapy
- Monitoring: Renal function, potassium levels (hypokalemia risk), thrombocytopenia
Option B: Pentavalent Antimonials (Sodium stibogluconate or Meglumine antimoniate)
- Dose: 20 mg/kg/day (IM or IV) for 30 days
- Caution: Requires cardiac monitoring (QT interval) due to risk of fatal arrhythmias and cardiotoxicity
- Contraindications to consider: None apparent in this young patient without mentioned cardiac/renal/hepatic disease
Alternative: Miltefosine or combination therapy if first-line fails.
3. Monitoring and Follow-up
- Clinical: Defervescence within 2–4 weeks, reduction in spleen size, weight gain
- Laboratory: Weekly CBC until pancytopenia resolves; repeat bone marrow aspirate if poor response
- Serological: rK39 may remain positive for months; not useful for cure monitoring
- Post-treatment surveillance: Monitor for Post-Kala-azar Dermal Leishmaniasis (PKDL) (hypopigmented macules/nodules appearing months to years later), which serves as a reservoir
Complications to Anticipate (Based on Pathogenesis)
- Severe bacterial infections: Pneumonia, septicemia (due to immune suppression and leukopenia)
- Hemorrhage: Epistaxis, GI bleeding (thrombocytopenia)
- Organ failure: Splenic rupture (rare but possible with massive splenomegaly)
- Relapse: Immunocompromised states (check HIV status)
Prognosis
Good if treated promptly. Without treatment, VL is universally fatal (>95% mortality). With appropriate therapy, mortality drops to <5%. Recovery of hematologic parameters typically occurs within 3–6 months.
Prevention Advice for Patient and Community
- Vector control: Sleep under insecticide-treated bed nets; indoor residual spraying with pyrethroids
- Early detection: Community awareness for similar symptoms (fever + abdominal swelling)
- Animal reservoirs: Control of stray dogs (major reservoir for L. infantum, though L. donovani is anthroponotic in Sudan)
Thank you